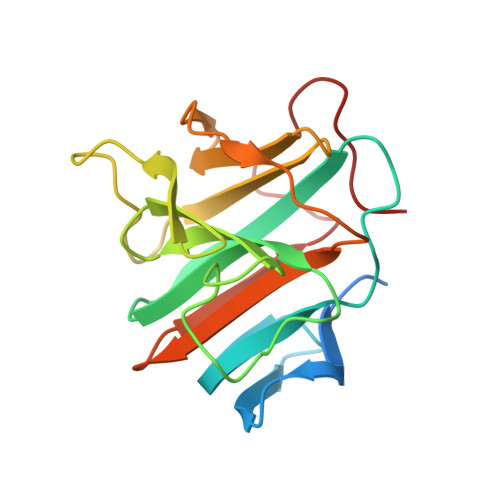
Crystal structure of the SPRY domain-containing protein 7 reveals unique structural features.
Yang, J., Guan, X., Zhang, D., Zhao, P., Guo, S., Kuang, Z.(2020) Biochem Biophys Res Commun 531: 350-356
- PubMed: 32800543
- DOI: https://doi.org/10.1016/j.bbrc.2020.07.076
- Primary Citation of Related Structures:
7CCB - PubMed Abstract:
The SPRY/B30.2 domain is one of the most abundant protein domains found in eukaryotes. Vast majority of the SPRY domain-containing proteins are multi-domain proteins. The SPRY domain-containing protein 7 (SPRY7, also named C13orf1, and named chronic lymphocytic leukemia deletion region gene 6 protein, CCLD6, encoded by the spryd7 gene) is the smallest SPRY domain protein in human that does not contain other accessory domains. Here we have determined the crystal structure of human SPRY7 at a resolution of 1.62 Å and found that SPRY7 has some unique structural features that are not present in other previously reported SRPY domain structures. Overall, SPRY7 may represent an evolutionary early version of the SPRY domain, and subsequent loop insertions and expansions, residue substitutions, as well as domain combinations have rendered the SPRY domain versatile binding specificities and broad biological functions. These results serve as a useful basis for a profound characterization of the molecular interactions of SPRY7.
Organizational Affiliation:
Department of Cell Biology, College of Life Science and Technology, Jinan University, Guangzhou, 510632, China; Institute of Biomedicine, Jinan University, Guangzhou, 510632, China; Guangdong Provincial Key Laboratory of Bioengineering Medicine, Guangzhou, 510632, China.