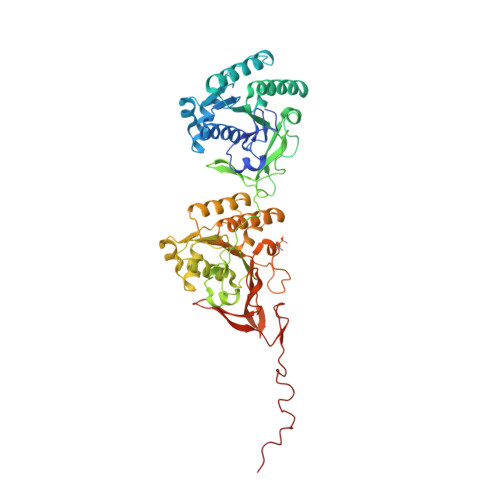
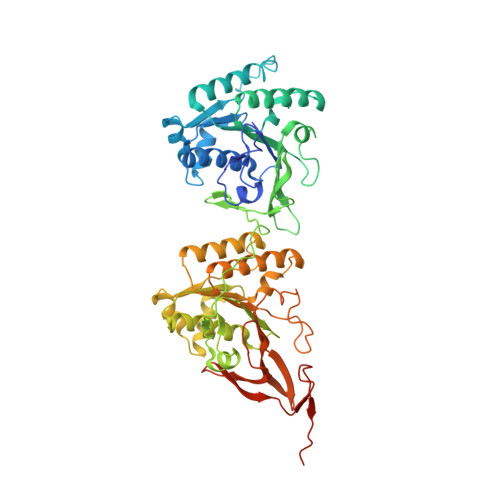
Structures of KaiC Circadian Clock Mutant Proteins: A New Phosphorylation Site at T426 and Mechanisms of Kinase, ATPase and Phosphatase.
Pattanayek, R., Mori, T., Xu, Y., Pattanayek, S., Johnson, C.H., Egli, M.(2009) PLoS One 4: e7529-e7529
- PubMed: 19956664
- DOI: https://doi.org/10.1371/journal.pone.0007529
- Primary Citation of Related Structures:
3JZM, 3K09, 3K0A, 3K0C, 3K0E, 3K0F - PubMed Abstract:
The circadian clock of the cyanobacterium Synechococcus elongatus can be reconstituted in vitro by three proteins, KaiA, KaiB and KaiC. Homo-hexameric KaiC displays kinase, phosphatase and ATPase activities; KaiA enhances KaiC phosphorylation and KaiB antagonizes KaiA. Phosphorylation and dephosphorylation of the two known sites in the C-terminal half of KaiC subunits, T432 and S431, follow a strict order (TS-->pTS-->pTpS-->TpS-->TS) over the daily cycle, the origin of which is not understood. To address this void and to analyze the roles of KaiC active site residues, in particular T426, we determined structures of single and double P-site mutants of S. elongatus KaiC.
Organizational Affiliation:
Department of Biochemistry, Vanderbilt University, School of Medicine, Nashville, Tennessee, United States of America.