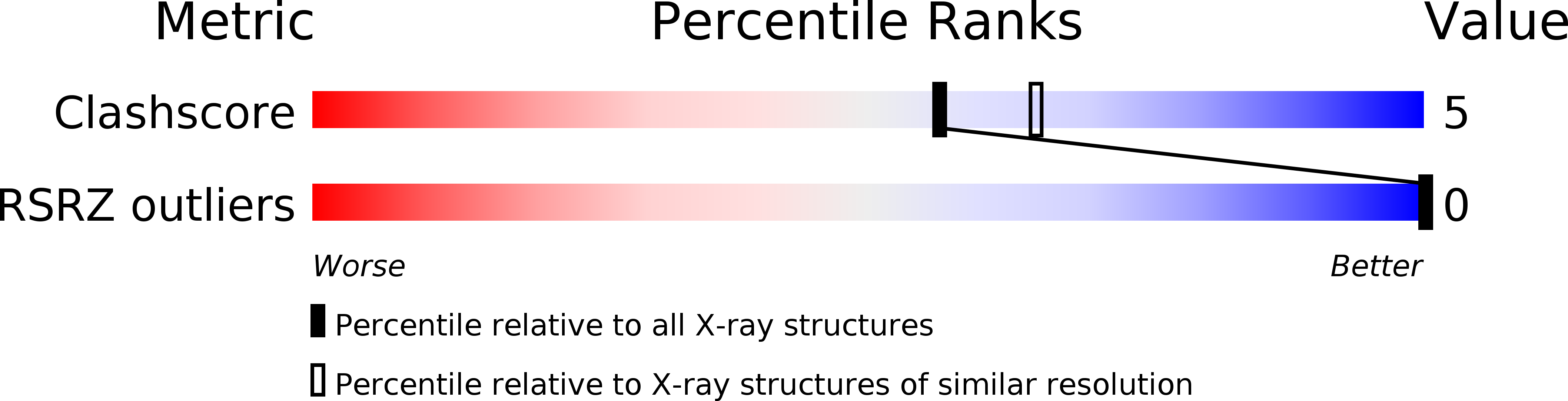A crystallographic and spectroscopic study of the complex between d(CGCGAATTCGCG)2 and 2,5-bis(4-guanylphenyl)furan, an analogue of berenil. Structural origins of enhanced DNA-binding affinity.
Laughton, C.A., Tanious, F., Nunn, C.M., Boykin, D.W., Wilson, W.D., Neidle, S.(1996) Biochemistry 35: 5655-5661
- PubMed: 8639524
- DOI: https://doi.org/10.1021/bi952162r
- Primary Citation of Related Structures:
227D - PubMed Abstract:
2,5-Bis(4-guanylphenyl)furan ("furamidine") is a dicationic minor groove binding drug that has been shown to be more effective than pentamidine against the Pneumocystis carinii pathogen in an immunosuppressed rat model. It has a close structural similarity to the antitrypanosomal drug berenil, differing only on the replacement of the central triazene unit with a furan moiety. we have determined the crystal structure of the complex between furamidine and the DNA dodecamer d(CGCGAATTCGCG)2 and compared it with the same DNA sequence by UV-visible, fluorescence, and CD spectroscopy. Furamidine shows tighter binding to this sequence (Keq = 6.7 x 10(6)) than berenil (Keq = 6.6 x 10(5)). The crystal structure reveals that, unlike berenil, furamidine makes direct hydrogen bond interactions with this DNA sequence through both amidinium groups to O2 atoms of thymine bases and is more isohelical with the minor groove. Molecular mechanics calculations support the hypothesis that these differences result in the improved interaction energy between the ligand and the DNA.
Organizational Affiliation:
CRC Biomolecular Structure Unit, Institute of Cancer Research, Sutton Surrey, UK.