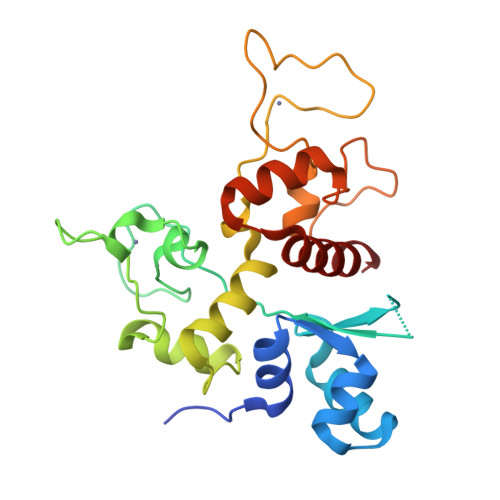
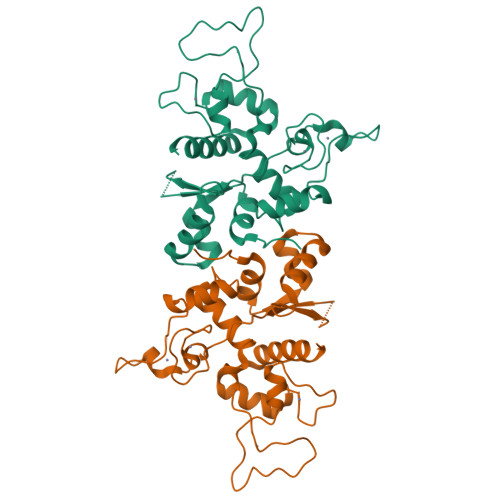
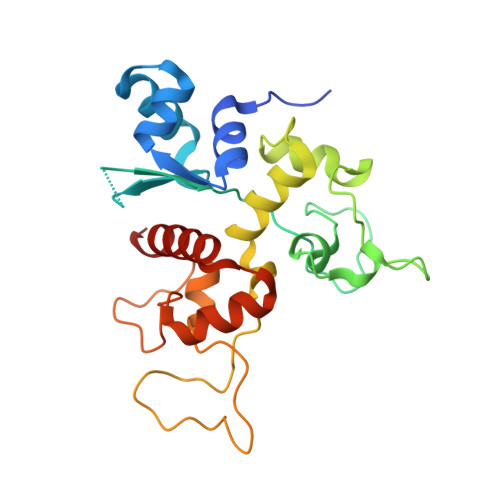
Structure of N-terminal domain of ZAP indicates how a zinc-finger protein recognizes complex RNA.
Chen, S., Xu, Y., Zhang, K., Wang, X., Sun, J., Gao, G., Liu, Y.(2012) Nat Struct Mol Biol 19: 430-435
- PubMed: 22407013
- DOI: https://doi.org/10.1038/nsmb.2243
- Primary Citation of Related Structures:
3U9G - PubMed Abstract:
Zinc-finger antiviral protein (ZAP) is a host factor that specifically inhibits the replication of certain viruses, such as HIV-1, by targeting viral mRNA for degradation. How ZAP recognizes its target RNA has been unclear. Here we report the crystal structure of the N-terminal domain of rat ZAP (NZAP225), the major functional domain. The overall structure of NZAP225 resembles a tractor, with four zinc-finger motifs located at the bottom. Structural and functional analyses identified multiple positively charged residues and two putative RNA-binding cavities forming a large putative RNA-binding cleft. ZAP molecules interact to form a dimer that binds to a ZAP-responsive RNA molecule containing two ZAP-binding modules. These results provide insights into how ZAP binds specifically to complex target RNA.
Organizational Affiliation:
State Key Laboratory of Biomacromolecules, Institute of Biophysics, Chinese Academy of Sciences, Beijing, China.