New Insights into the Assembly of Bacterial Secretins: STRUCTURAL STUDIES OF THE PERIPLASMIC DOMAIN OF XcpQ FROM PSEUDOMONAS AERUGINOSA.
Van der Meeren, R., Wen, Y., Van Gelder, P., Tommassen, J., Devreese, B., Savvides, S.N.(2013) J Biological Chem 288: 1214-1225
- PubMed: 23188826
- DOI: https://doi.org/10.1074/jbc.M112.432096
- Primary Citation of Related Structures:
4E9J, 4EC5 - PubMed Abstract:
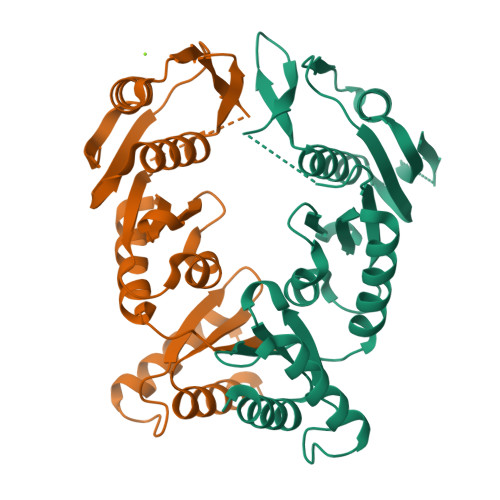
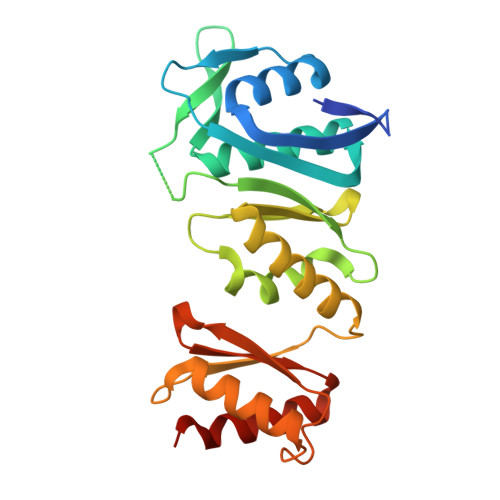
The type II secretion system is a multiprotein assembly spanning the inner and outer membranes in Gram-negative bacteria. It is found in almost all pathogenic bacteria where it contributes to virulence, host tissue colonization, and infection. The exoproteins are secreted across the outer membrane via a large translocation channel, the secretin, which typically adopts a dodecameric structure. These secretin channels have large periplasmic N-terminal domains that reach out into the periplasm for communication with the inner membrane platform and with a pseudopilus structure that spans the periplasm. Here we report the crystal structure of the N-terminal periplasmic domain of the secretin XcpQ from Pseudomonas aeruginosa, revealing a two-lobe dimeric assembly featuring parallel subunits engaging in well defined interactions at the tips of each lobe. We have employed structure-based engineering of disulfide bridges and native mass spectrometry to show that the periplasmic domain of XcpQ dimerizes in a concentration-dependent manner. Validation of these insights in the context of cellular full-length XcpQ and further evaluation of the functionality of disulfide-linked XcpQ establishes that the basic oligomerization unit of XcpQ is a dimer. This is consistent with the notion that the dodecameric secretin assembles as a hexamer of dimers to ensure correct projection of the N-terminal domains into the periplasm. Therefore, our studies provide a key conceptual advancement in understanding the assembly principles and dynamic function of type II secretion system secretins and challenge recent studies reporting monomers as the basic subunit of the secretin oligomer.
Organizational Affiliation:
Unit for Structural Biology, Ghent University, 9000 Ghent, Belgium.