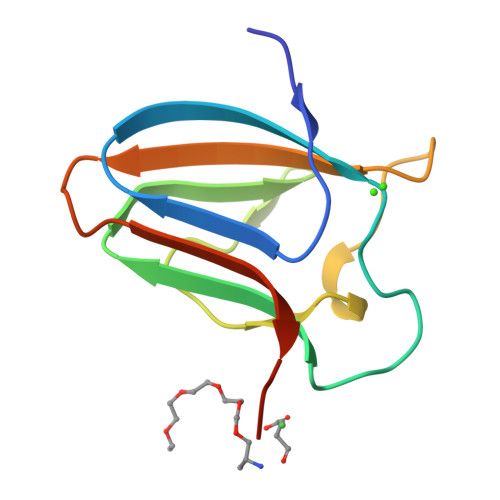
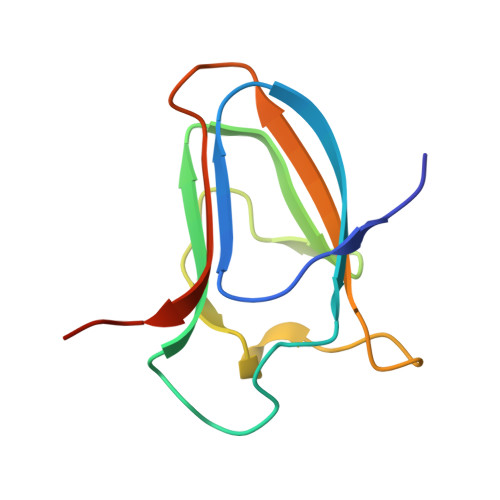
Stability and Ligand Promiscuity of Type A Carbohydrate-binding Modules Are Illustrated by the Structure of Spirochaeta thermophila StCBM64C.
Pires, V.M., Pereira, P.M., Bras, J.L., Correia, M., Cardoso, V., Bule, P., Alves, V.D., Najmudin, S., Venditto, I., Ferreira, L.M., Romao, M.J., Carvalho, A.L., Fontes, C.M., Prazeres, D.M.(2017) J Biological Chem 292: 4847-4860
- PubMed: 28179427
- DOI: https://doi.org/10.1074/jbc.M116.767541
- Primary Citation of Related Structures:
5LU3 - PubMed Abstract:
Deconstruction of cellulose, the most abundant plant cell wall polysaccharide, requires the cooperative activity of a large repertoire of microbial enzymes. Modular cellulases contain non-catalytic type A carbohydrate-binding modules (CBMs) that specifically bind to the crystalline regions of cellulose, thus promoting enzyme efficacy through proximity and targeting effects. Although type A CBMs play a critical role in cellulose recycling, their mechanism of action remains poorly understood. Here we produced a library of recombinant CBMs representative of the known diversity of type A modules. The binding properties of 40 CBMs, in fusion with an N-terminal GFP domain, revealed that type A CBMs possess the ability to recognize different crystalline forms of cellulose and chitin over a wide range of temperatures, pH levels, and ionic strengths. A Spirochaeta thermophila CBM64, in particular, displayed plasticity in its capacity to bind both crystalline and soluble carbohydrates under a wide range of extreme conditions. The structure of S. thermophila St CBM64C revealed an untwisted, flat, carbohydrate-binding interface comprising the side chains of four tryptophan residues in a co-planar linear arrangement. Significantly, two highly conserved asparagine side chains, each one located between two tryptophan residues, are critical to insoluble and soluble glucan recognition but not to bind xyloglucan. Thus, CBM64 compact structure and its extended and versatile ligand interacting platform illustrate how type A CBMs target their appended plant cell wall-degrading enzymes to a diversity of recalcitrant carbohydrates under a wide range of environmental conditions.
Organizational Affiliation:
From the CIISA, Faculdade de Medicina Veterinária, Universidade de Lisboa, Pólo Universitário da Ajuda, 1300-477 Lisboa, Portugal.