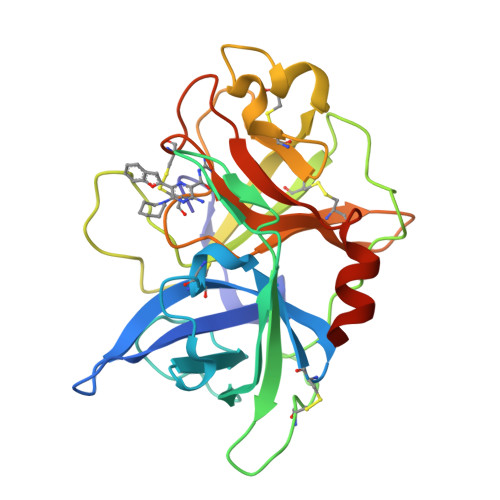
6-Substituted Hexamethylene Amiloride (HMA) Derivatives as Potent and Selective Inhibitors of the Human Urokinase Plasminogen Activator for Use in Cancer.
Buckley, B.J., Aboelela, A., Minaei, E., Jiang, L.X., Xu, Z., Ali, U., Fildes, K., Cheung, C.Y., Cook, S.M., Johnson, D.C., Bachovchin, D.A., Cook, G.M., Apte, M., Huang, M., Ranson, M., Kelso, M.J.(2018) J Med Chem 61: 8299-8320
- PubMed: 30130401
- DOI: https://doi.org/10.1021/acs.jmedchem.8b00838
- Primary Citation of Related Structures:
5ZA7, 5ZA8, 5ZA9, 5ZAE, 5ZAF, 5ZAG, 5ZAH, 5ZAJ, 5ZC5 - PubMed Abstract:
Metastasis is the cause of death in the majority (∼90%) of malignant cancers. The oral potassium-sparing diuretic amiloride and its 5-substituted derivative 5 -N, N-(hexamethylene)amiloride (HMA) reportedly show robust antitumor/metastasis effects in multiple in vitro and animal models. These effects are likely due, at least in part, to inhibition of the urokinase plasminogen activator (uPA), a key protease determinant of cell invasiveness and metastasis. This study reports the discovery of 6-substituted HMA analogs that show nanomolar potency against uPA, high selectivity over related trypsin-like serine proteases, and minimal inhibitory effects against epithelial sodium channels (ENaC), the diuretic and antikaliuretic target of amiloride. Reductions in lung metastases were demonstrated for two analogs in a late-stage experimental mouse metastasis model, and one analog completely inhibited formation of liver metastases in an orthotopic xenograft mouse model of pancreatic cancer. The results support further evaluation of 6-substituted HMA derivatives as uPA-targeting anticancer drugs.
Organizational Affiliation:
Molecular Horizons and School of Chemistry & Molecular Bioscience , University of Wollongong , Wollongong , NSW 2522 , Australia.