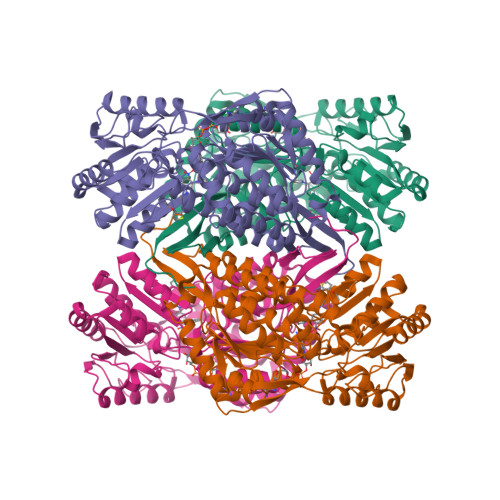
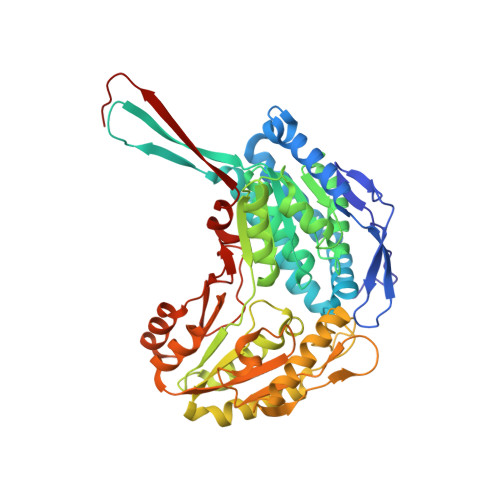
Structural Basis of ALDH1A2 Inhibition by Irreversible and Reversible Small Molecule Inhibitors.
Chen, Y., Zhu, J.Y., Hong, K.H., Mikles, D.C., Georg, G.I., Goldstein, A.S., Amory, J.K., Schonbrunn, E.(2018) ACS Chem Biol 13: 582-590
- PubMed: 29240402
- DOI: https://doi.org/10.1021/acschembio.7b00685
- Primary Citation of Related Structures:
6ALJ, 6B5G, 6B5H, 6B5I - PubMed Abstract:
Enzymes of the ALDH1A subfamily of aldehyde dehydrogenases are crucial in regulating retinoic acid (RA) signaling and have received attention as potential drug targets. ALDH1A2 is the primary RA-synthesizing enzyme in mammalian spermatogenesis and is therefore considered a viable drug target for male contraceptive development. However, only a small number of ALDH1A2 inhibitors have been reported, and information on the structure of ALDH1A2 was limited to the NAD-liganded enzyme void of substrate or inhibitors. Herein, we describe the mechanism of action of structurally unrelated reversible and irreversible inhibitors of human ALDH1A2 using direct binding studies and X-ray crystallography. All inhibitors bind to the active sites of tetrameric ALDH1A2. Compound WIN18,446 covalently reacts with the side chain of the catalytic residue Cys320, resulting in a chiral adduct in ( R) configuration. The covalent adduct directly affects the neighboring NAD molecule, which assumes a contracted conformation suboptimal for the dehydrogenase reaction. The reversible inhibitors interact predominantly through direct hydrogen bonding interactions with residues in the vicinity of Cys320 without affecting NAD. Upon interaction with inhibitors, a large flexible loop assumes regular structure, thereby shielding the active site from solvent. The precise knowledge of the binding modes provides a new framework for the rational design of novel inhibitors of ALDH1A2 with improved potency and selectivity profiles.
Organizational Affiliation:
Drug Discovery Department , Moffitt Cancer Center , Tampa , Florida 33612 , United States.