Multicomponent Peptide Stapling as a Diversity-Driven Tool for the Development of Inhibitors of Protein-Protein Interactions.
Ricardo, M.G., Ali, A.M., Plewka, J., Surmiak, E., Labuzek, B., Neochoritis, C.G., Atmaj, J., Skalniak, L., Zhang, R., Holak, T.A., Groves, M., Rivera, D.G., Domling, A.(2020) Angew Chem Int Ed Engl 59: 5235-5241
- PubMed: 31944488
- DOI: https://doi.org/10.1002/anie.201916257
- Primary Citation of Related Structures:
6T2D, 6T2E, 6T2F - PubMed Abstract:
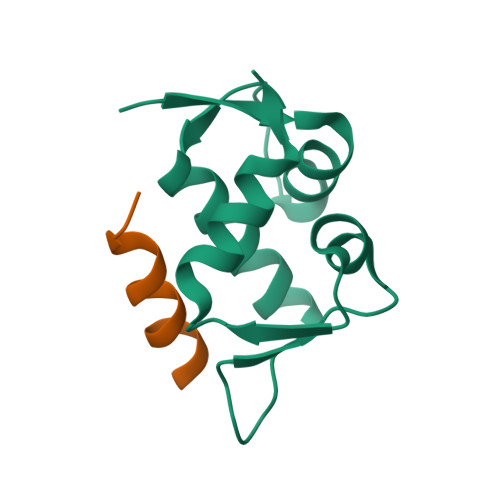
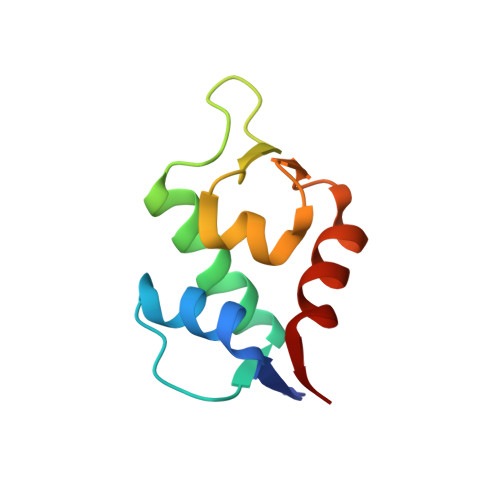
Stapled peptides are chemical entities in-between biologics and small molecules, which have proven to be the solution to high affinity protein-protein interaction antagonism, while keeping control over pharmacological performance such as stability and membrane penetration. We demonstrate that the multicomponent reaction-based stapling is an effective strategy for the development of α-helical peptides with highly potent dual antagonistic action of MDM2 and MDMX binding p53. Such a potent inhibitory activity of p53-MDM2/X interactions was assessed by fluorescence polarization, microscale thermophoresis, and 2D NMR, while several cocrystal structures with MDM2 were obtained. This MCR stapling protocol proved efficient and versatile in terms of diversity generation at the staple, as evidenced by the incorporation of both exo- and endo-cyclic hydrophobic moieties at the side chain cross-linkers. The interaction of the Ugi-staple fragments with the target protein was demonstrated by crystallography.
Organizational Affiliation:
Faculty of Chemistry, Center for Natural Product Research, University of Havana, Cuba.