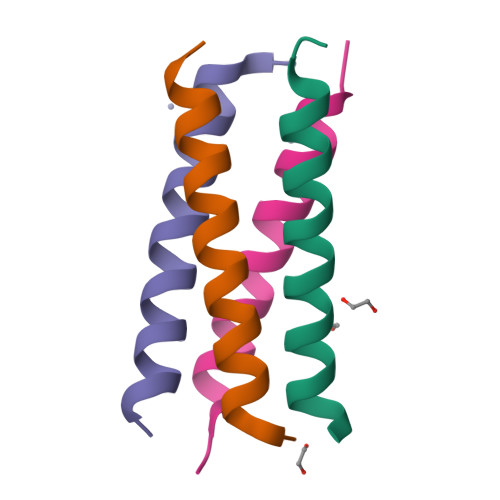
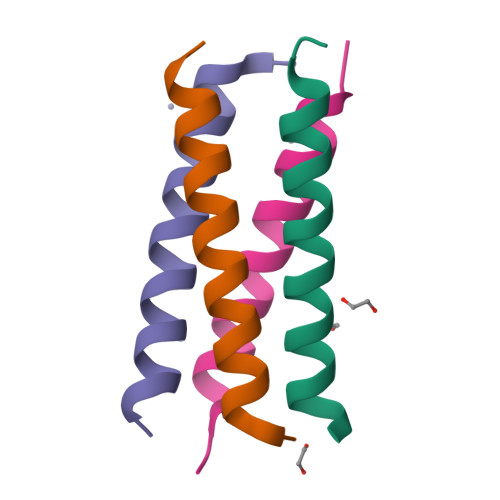
Structural polymorphism of coiled-coils from the stalk domain of SARS-CoV-2 spike protein.
Zivic, Z., Strmsek, Z., Novinec, M., Lah, J., Hadzi, S.(2022) FASEB J 36: e22199-e22199
- PubMed: 35157347
- DOI: https://doi.org/10.1096/fj.202101670R
- Primary Citation of Related Structures:
7OWX - PubMed Abstract:
Spike trimer plays a key role in SARS-CoV-2 infection and vaccine development. It consists of a globular head and a flexible stalk domain that anchors the protein into the viral membrane. While the head domain has been extensively studied, the properties of the adjoining stalk are poorly understood. Here, we characterize the coiled-coil formation and thermodynamic stability of the stalk domain and its segments. We find that the N-terminal segment of the stalk does not form coiled-coils and remains disordered in solution. The C-terminal stalk segment forms a trimeric coiled-coil in solution, which becomes significantly stabilized in the context of the full-length stalk. Its crystal structure reveals a novel antiparallel tetramer coiled-coil with an unusual combination of a-d and e-a-d hydrophobic core packing. Structural analysis shows that a subset of hydrophobic residues stabilizes different coiled-coil structures: trimer, tetramer, and heterohexamer, underscoring a highly polymorphic nature of the SARS-CoV-2 stalk sequence.
Organizational Affiliation:
Faculty of Chemistry and Chemical Technology, University of Ljubljana, Ljubljana, Slovenia.