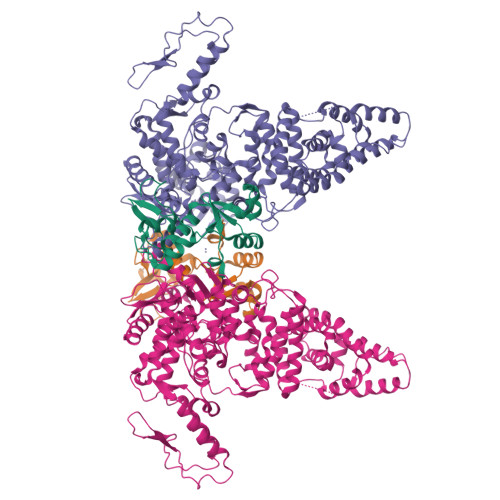
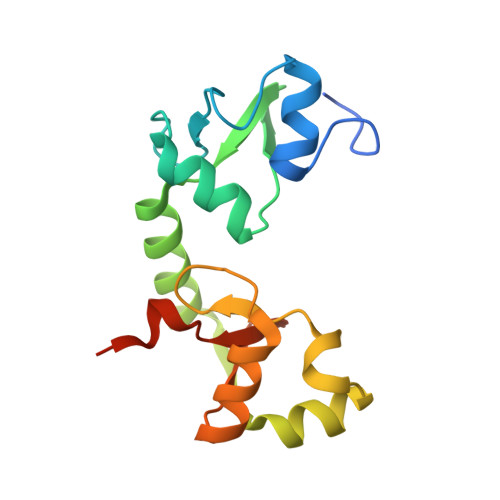
Structural insights into the functional mechanism of the ubiquitin ligase E6AP.
Wang, Z., Fan, F., Li, Z., Ye, F., Wang, Q., Gao, R., Qiu, J., Lv, Y., Lin, M., Xu, W., Luo, C., Yu, X.(2024) Nat Commun 15: 3531-3531
- PubMed: 38670961
- DOI: https://doi.org/10.1038/s41467-024-47586-w
- Primary Citation of Related Structures:
8JRN, 8JRO, 8JRP, 8JRQ, 8JRR - PubMed Abstract:
E6AP dysfunction is associated with Angelman syndrome and Autism spectrum disorder. Additionally, the host E6AP is hijacked by the high-risk HPV E6 to aberrantly ubiquitinate the tumor suppressor p53, which is linked with development of multiple types of cancer, including most cervical cancers. Here we show that E6AP and the E6AP/E6 complex exist, respectively, as a monomer and a dimer of the E6AP/E6 protomer. The short α1-helix of E6AP transforms into a longer helical structure when in complex with E6. The extended α1-helices of the dimer intersect symmetrically and contribute to the dimerization. The two protomers sway around the crossed region of the two α1-helices to promote the attachment and detachment of substrates to the catalytic C-lobe of E6AP, thus facilitating ubiquitin transfer. These findings, complemented by mutagenesis analysis, suggest that the α1-helix, through conformational transformations, controls the transition between the inactive monomer and the active dimer of E6AP.
Organizational Affiliation:
State Key Laboratory of Drug Research, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, Shanghai, 201203, China.