X-ray crystallographic studies of recombinant inorganic pyrophosphatase from Escherichia coli.
Oganessyan, V.Y.u., Kurilova, S.A., Vorobyeva, N.N., Nazarova, T.I., Popov, A.N., Lebedev, A.A., Avaeva, S.M., Harutyunyan, E.H.(1994) FEBS Lett 348: 301-304
- PubMed: 8034059
- DOI: https://doi.org/10.1016/0014-5793(94)00605-9
- Primary Citation of Related Structures:
1IGP - PubMed Abstract:
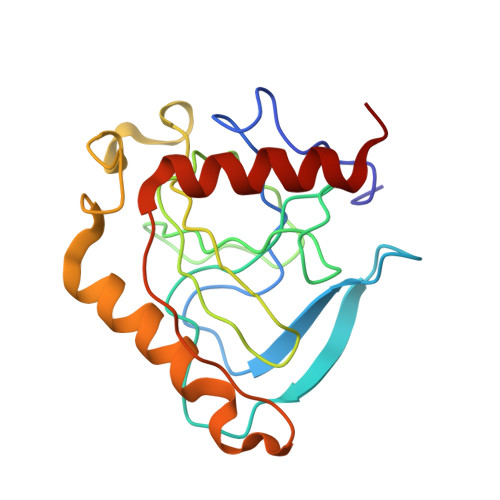
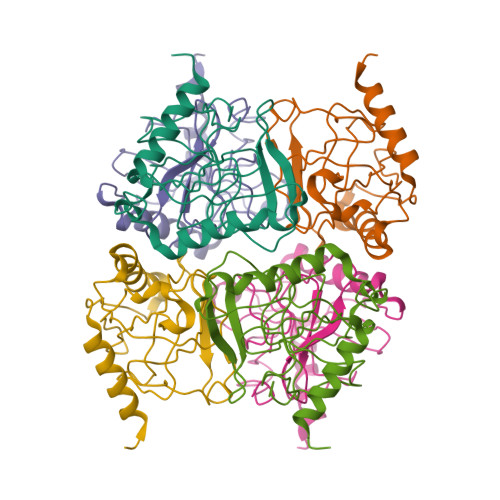
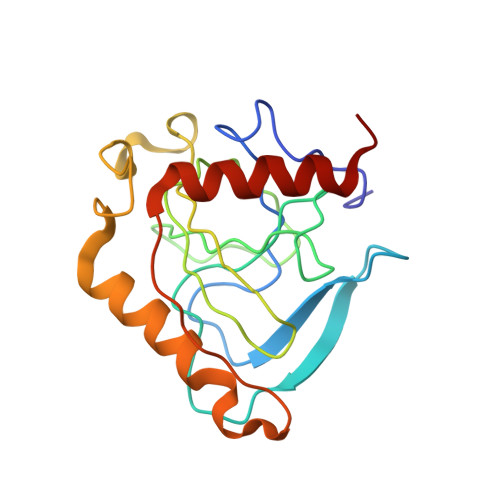
An E. coli inorganic pyrophosphatase overproducer and a method for a large-scale production of the homogeneous enzyme are described. The inorganic pyrophosphatase was crystallized in the form containing one subunit of a homohexameric molecule per asymmetric unit: space group R32, a = 110.4 A, c = 76.8 A. The electron density map to 2.5 A resolution phased with Eu- and Hg-derivatives (figure of merit,
= 0.51) was improved by the solvent flattening procedure ( = 0.77). The course of the polypeptide chain and the secondary structure elements, intersubunit contacts and positions of the active sites were characterized. Homology with S. cerevisiae inorganic pyrophosphatase structure was found.
Organizational Affiliation:
Institute of Crystallography, Russian Academy of Sciences, Moscow.