Origin of exopolyphosphatase processivity: Fusion of an ASKHA phosphotransferase and a cyclic nucleotide phosphodiesterase homolog.
Alvarado, J., Ghosh, A., Janovitz, T., Jauregui, A., Hasson, M.S., Sanders, D.A.(2006) Structure 14: 1263-1272
- PubMed: 16905100
- DOI: https://doi.org/10.1016/j.str.2006.06.009
- Primary Citation of Related Structures:
1U6Z - PubMed Abstract:
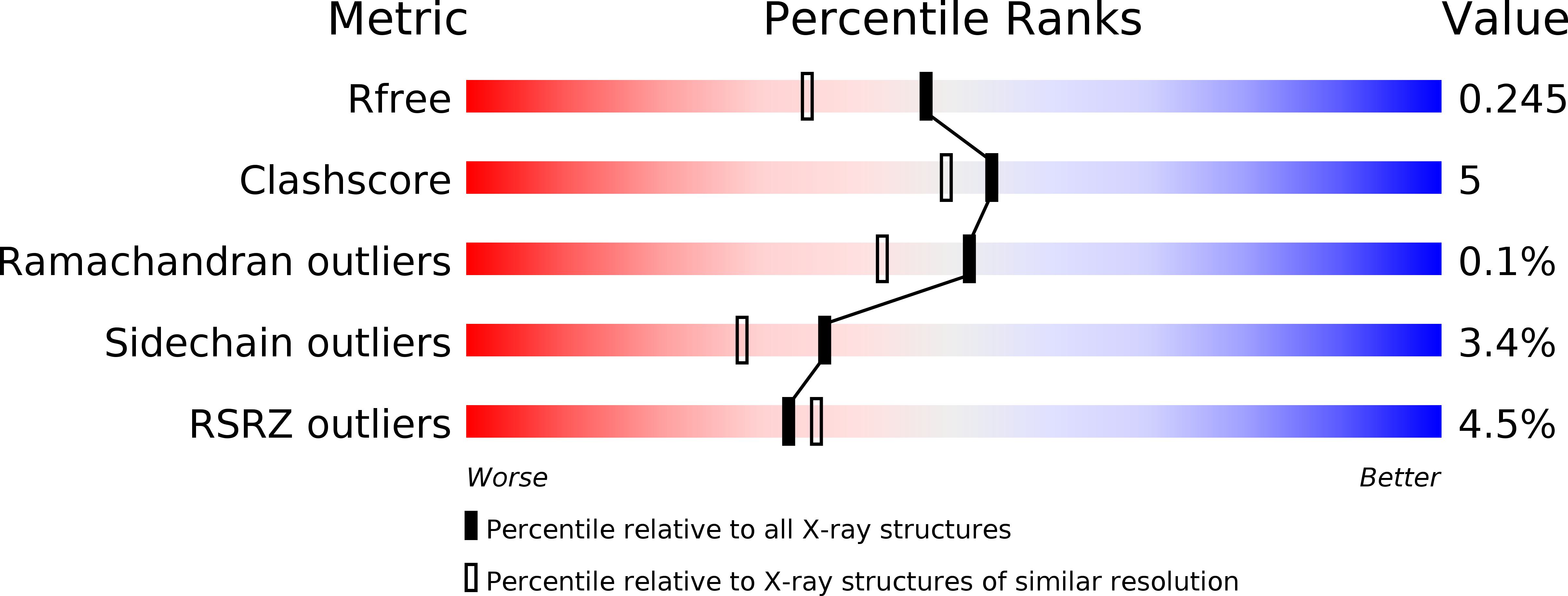
The Escherichia coli Ppx protein is an exopolyphosphatase that degrades long-chain polyphosphates in a highly processive reaction. It also hydrolyzes the terminal 5' phosphate of the modified nucleotide guanosine 5' triphosphate 3' diphosphate (pppGpp). The structure of Ppx has been determined to 1.9 A resolution by X-ray crystallography. The exopolyphosphatase is an ASKHA (acetate and sugar kinases, Hsp70, actin) phosphotransferase with an active site found in a cleft between the two amino-terminal domains. Analysis of the active site indicates that among the ASKHA phosphotranferases of known structure, Ppx is the closest to the ectonucleoside triphosphate diphosphohydrolases. A third domain forms a six-helix claw that is similar to the catalytic core of the eukaryotic cyclic nucleotide phosphodiesterases. Most of the 29 sulfate ions bound to the Ppx dimer occupy sites where the polyP chain likely binds. An aqueduct that passes through the enzyme provides a physical basis for the enzyme's high processivity.
Organizational Affiliation:
Department of Biological Sciences, Purdue University, West Lafayette, Indiana 47907, USA.