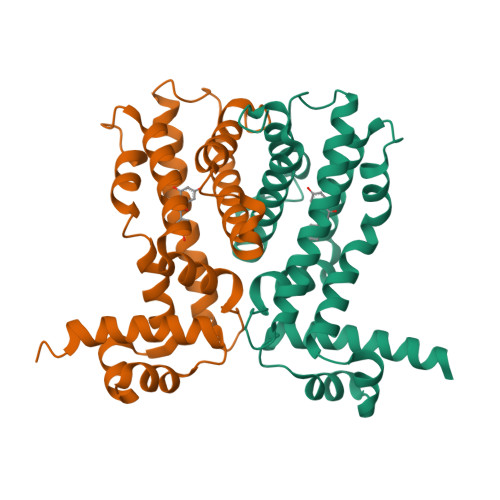
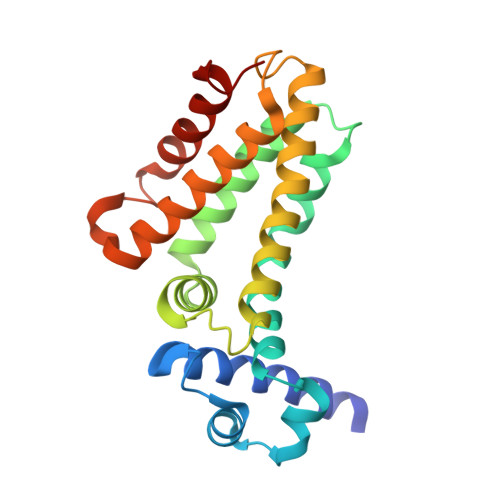
Crystal Structures of Multidrug Binding Protein Ttgr in Complex with Antibiotics and Plant Antimicrobials.
Alguel, Y., Meng, C., Teran, W., Krell, T., Ramos, J.L., Gallegos, M.-T., Zhang, X.(2007) J Mol Biology 369: 829
- PubMed: 17466326
- DOI: https://doi.org/10.1016/j.jmb.2007.03.062
- Primary Citation of Related Structures:
2UXH, 2UXI, 2UXO, 2UXP, 2UXU - PubMed Abstract:
Antibiotic resistance is a widely spread phenomenon. One major mechanism that underlies antibiotic resistance in bacteria is the active extrusion of toxic compounds through the membrane-bound efflux pumps that are often regulated at the transcriptional level. TtgR represses the transcription of TtgABC, a key efflux pump in Pseudomonas putida, which is highly resistant to antibiotics, solvents and toxic plant secondary products. Previously we showed that TtgR is the only reported repressor that binds to different classes of natural antimicrobial compounds, which are also extruded by the efflux pump. We report here five high-resolution crystal structures of TtgR from the solvent-tolerant strain DOT-T1E, including TtgR in complex with common antibiotics and plant secondary metabolites. We provide structural basis for the unique ligand binding properties of TtgR. We identify two distinct and overlapping ligand binding sites; the first one is broader and consists of mainly hydrophobic residues, whereas the second one is deeper and contains more polar residues including Arg176, a unique residue present in the DOT-T1E strain but not in other Pseudomonas strains. Phloretin, a plant antimicrobial, can bind to both binding sites with distinct binding affinities and stoichiometries. Results on ligand binding properties of native and mutant TtgR proteins using isothermal titration calorimetry confirm the binding affinities and stoichiometries, and suggest a potential positive cooperativity between the two binding sites. The importance of Arg176 in phloretin binding was further confirmed by the reduced ability of phloretin in releasing the mutant TtgR from bound DNA compared to the native protein. The results presented here highlight the importance and versatility of regulatory systems in bacterial antibiotic resistance and open up new avenues for novel antimicrobial development.
Organizational Affiliation:
Centre for Structural Biology and Division of Molecular Biosciences, Imperial College London, London, SW7 2AZ, UK.