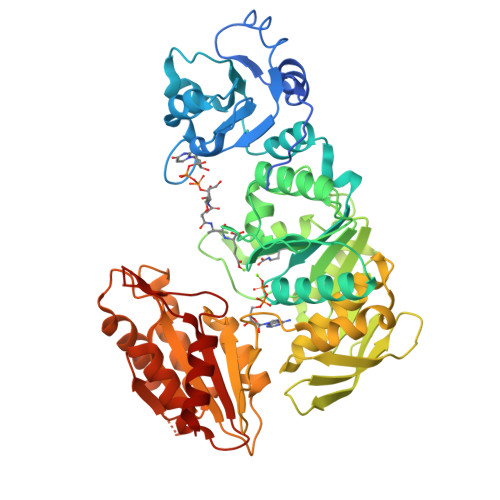
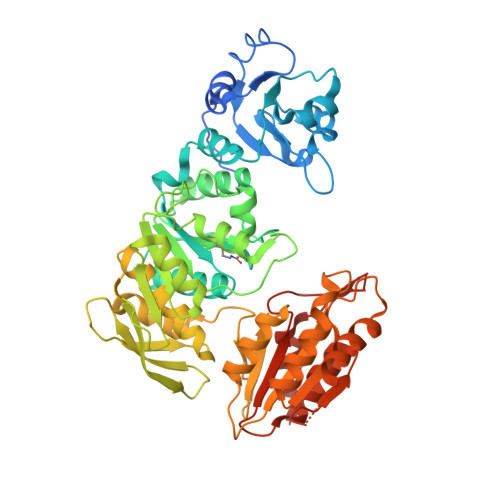
Essential Residues for the Enzyme Activity of ATP-Dependent Mure Ligase from Mycobacterium Tuberculosis.
Basavannacharya, C., Moody, P.R., Munshi, T., Cronin, N., Keep, N.H., Bhakta, S.(2010) Protein Cell 1: 1011
- PubMed: 21153518
- DOI: https://doi.org/10.1007/s13238-010-0132-9
- Primary Citation of Related Structures:
2XJA - PubMed Abstract:
The emergence of total drug-resistant tuberculosis (TDRTB) has made the discovery of new therapies for tuberculosis urgent. The cytoplasmic enzymes of peptidoglycan biosynthesis have generated renewed interest as attractive targets for the development of new anti-mycobacterials. One of the cytoplasmic enzymes, uridine diphosphate (UDP)-MurNAc-tripeptide ligase (MurE), catalyses the addition of meso-diaminopimelic acid (m-DAP) into peptidoglycan in Mycobacterium tuberculosis coupled to the hydrolysis of ATP. Mutants of M. tuberculosis MurE were generated by replacing K157, E220, D392, R451 with alanine and N449 with aspartate, and truncating the first 24 amino acid residues at the N-terminus of the enzyme. Analysis of the specific activity of these proteins suggested that apart from the 24 N-terminal residues, the other mutated residues are essential for catalysis. Variations in K(m) values for one or more substrates were observed for all mutants, except the N-terminal truncation mutant, indicating that these residues are involved in binding substrates and form part of the active site structure. These mutant proteins were also tested for their specificity for a wide range of substrates. Interestingly, the mutations K157A, E220A and D392A showed hydrolysis of ATP uncoupled from catalysis. The ATP hydrolysis rate was enhanced by at least partial occupation of the uridine nucleotide dipeptide binding site. This study provides an insight into the residues essential for the catalytic activity and substrate binding of the ATP-dependent MurE ligase. Since ATP-dependent MurE ligase is a novel drug target, the understanding of its function may lead to development of novel inhibitors against resistant forms of M. tuberculosis.
Organizational Affiliation:
Department of Biological Sciences, Institute for Structural and Molecular Biology, Birkbeck, UK.