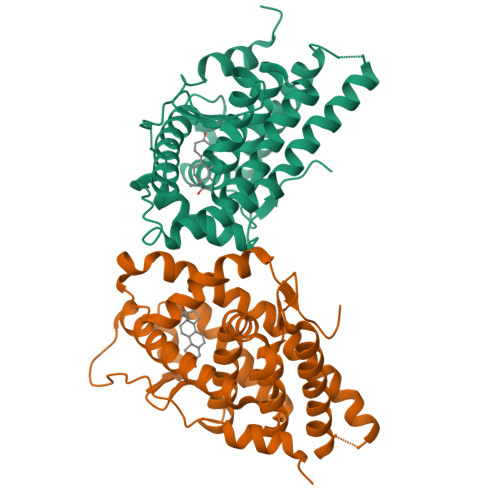
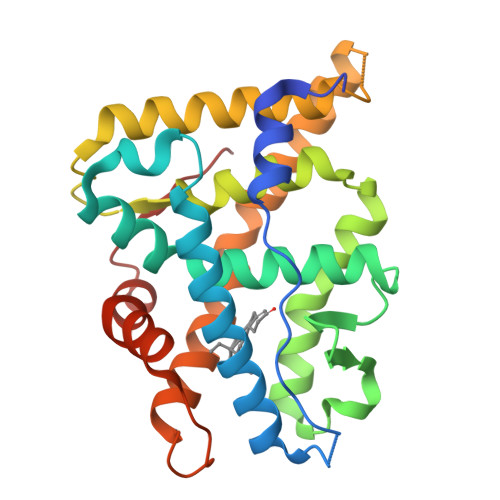
Met909 plays a key role in the activation of the progesterone receptor and also in the high potency of 13-ethyl progestins
Petit-Topin, I., Turque, N., Fagart, J., Fay, M., Ulmann, A., Gainer, E., Rafestin-Oblin, M.E.(2009) Mol Pharmacol 75: 1317-1324
- PubMed: 19289570
- DOI: https://doi.org/10.1124/mol.108.054312
- Primary Citation of Related Structures:
3D90 - PubMed Abstract:
Many progestins have been developed for use in contraception, menopausal hormone therapy, and treatment of gynecological diseases. They are derived from either progesterone or testosterone, and they act by binding to the progesterone receptor (PR), a hormone-inducible transcription factor belonging to the nuclear receptor superfamily. Unlike mineralocorticoid, glucocorticoid, and androgen receptors, the steroid-receptor contacts that trigger the switch of the ligand-binding domain from an inactive to an active conformation have not yet been identified for the PR. With this aim, we solved the crystal structure of the ligand-binding domain of the human PR complexed with levonorgestrel, a potent testosterone-derived progestin characterized by a 13-ethyl substituent. Via mutagenesis analysis and functional studies, we identified Met909 of the helix 12 as the key residue for PR activation by both testosterone- and progesterone-derived progestins with a 13-methyl or a 13-ethyl substituent. We also showed that Asn719 contributes to PR activation by testosterone-derived progestins only, and that Met759 and Met909 are responsible for the high potency of 19-norprogestins and of 13-ethyl progestins, respectively. Our findings provide a structural guideline for the rational synthesis of potent PR agonist and antagonist ligands that could have therapeutic uses in women's health.
Organizational Affiliation:
INSERM U, Centre de Recherche Biomédicale Bichat-Beaujon, Paris, France.