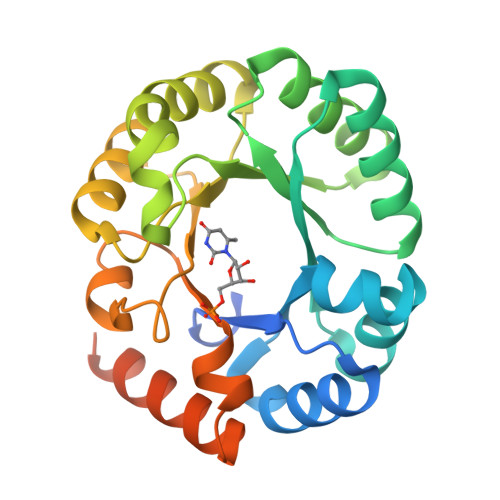
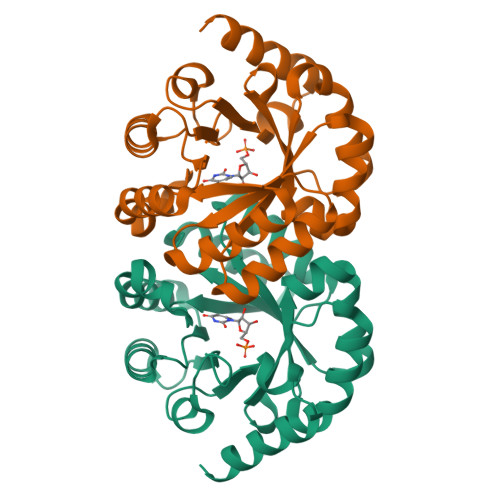
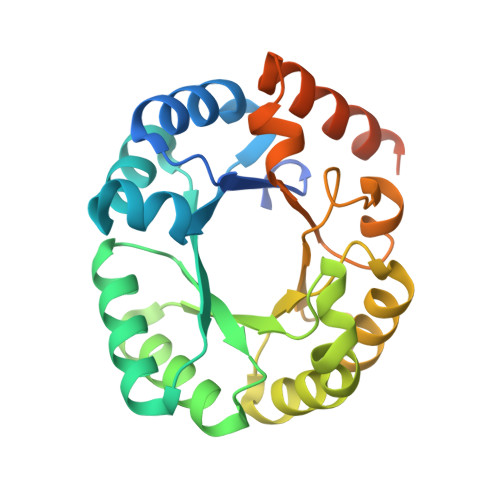
Substrate distortion contributes to the catalysis of orotidine 5'-monophosphate decarboxylase.
Fujihashi, M., Ishida, T., Kuroda, S., Kotra, L.P., Pai, E.F., Miki, K.(2013) J Am Chem Soc 135: 17432-17443
- PubMed: 24151964
- DOI: https://doi.org/10.1021/ja408197k
- Primary Citation of Related Structures:
3WJW, 3WJX, 3WJY, 3WJZ, 3WK0, 3WK1, 3WK2, 3WK3 - PubMed Abstract:
Orotidine 5'-monophosphate decarboxylase (ODCase) accelerates the decarboxylation of orotidine 5'-monophosphate (OMP) to uridine 5'-monophosphate (UMP) by 17 orders of magnitude. Eight new crystal structures with ligand analogues combined with computational analyses of the enzyme's short-lived intermediates and the intrinsic electronic energies to distort the substrate and other ligands improve our understanding of the still controversially discussed reaction mechanism. In their respective complexes, 6-methyl-UMP displays significant distortion of its methyl substituent bond, 6-amino-UMP shows the competition between the K72 and C6 substituents for a position close to D70, and the methyl and ethyl esters of OMP both induce rotation of the carboxylate group substituent out of the plane of the pyrimidine ring. Molecular dynamics and quantum mechanics/molecular mechanics computations of the enzyme-substrate complex also show the bond between the carboxylate group and the pyrimidine ring to be distorted, with the distortion contributing a 10-15% decrease of the ΔΔG(⧧) value. These results are consistent with ODCase using both substrate distortion and transition-state stabilization, primarily exerted by K72, in its catalysis of the OMP decarboxylation reaction.
Organizational Affiliation:
Department of Chemistry, Graduate School of Science, Kyoto University , Sakyo-ku, Kyoto 606-8502, Japan.