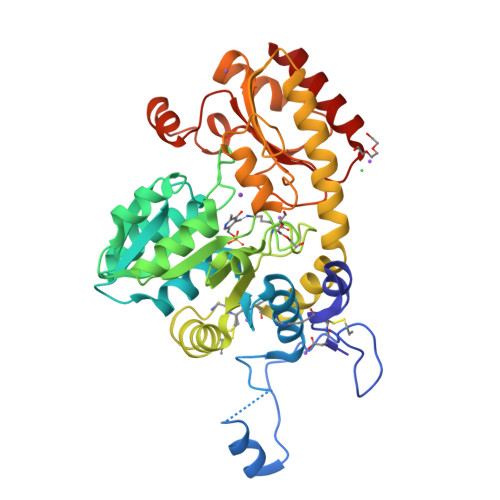
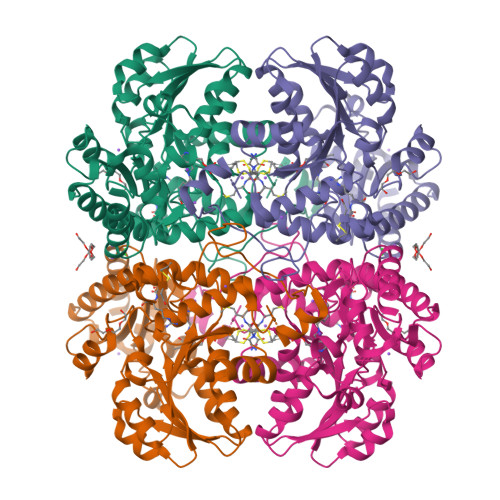
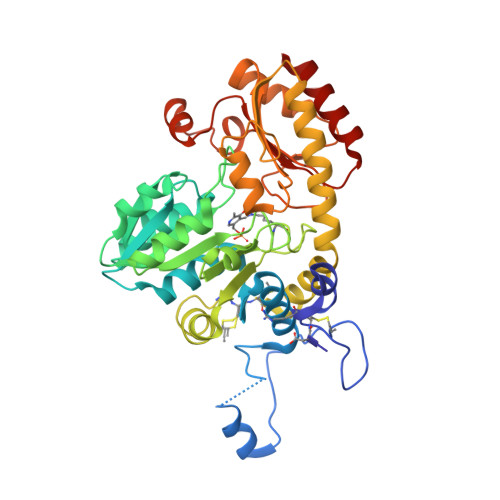
Alliin is a suicide substrate of Citrobacter freundii methionine gamma-lyase: structural bases of inactivation of the enzyme.
Morozova, E.A., Revtovich, S.V., Anufrieva, N.V., Kulikova, V.V., Nikulin, A.D., Demidkina, T.V.(2014) Acta Crystallogr D Biol Crystallogr 70: 3034-3042
- PubMed: 25372692
- DOI: https://doi.org/10.1107/S1399004714020938
- Primary Citation of Related Structures:
4MKJ, 4MKK - PubMed Abstract:
The interaction of Citrobacter freundii methionine γ-lyase (MGL) and the mutant form in which Cys115 is replaced by Ala (MGL C115A) with the nonprotein amino acid (2R)-2-amino-3-[(S)-prop-2-enylsulfinyl]propanoic acid (alliin) was investigated. It was found that MGL catalyzes the β-elimination reaction of alliin to form 2-propenethiosulfinate (allicin), pyruvate and ammonia. The β-elimination reaction of alliin is followed by the inactivation and modification of SH groups of the wild-type and mutant enzymes. Three-dimensional structures of inactivated wild-type MGL (iMGL wild type) and a C115A mutant form (iMGL C115A) were determined at 1.85 and 1.45 Å resolution and allowed the identification of the SH groups that were oxidized by allicin. On this basis, the mechanism of the inactivation of MGL by alliin, a new suicide substrate of MGL, is proposed.
Organizational Affiliation:
Engelhardt Institute of Molecular Biology, Russian Academy of Science, Vavilov Str. 32, Moscow 119991, Russian Federation.