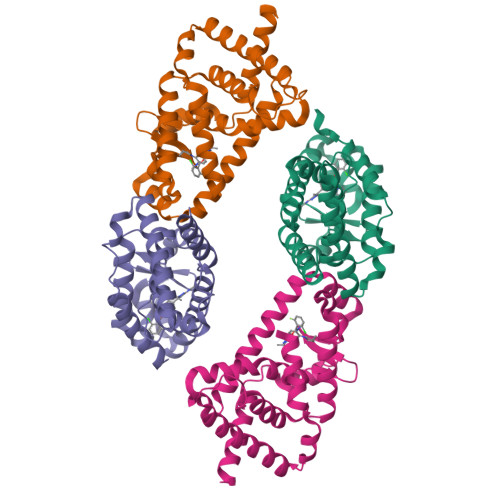
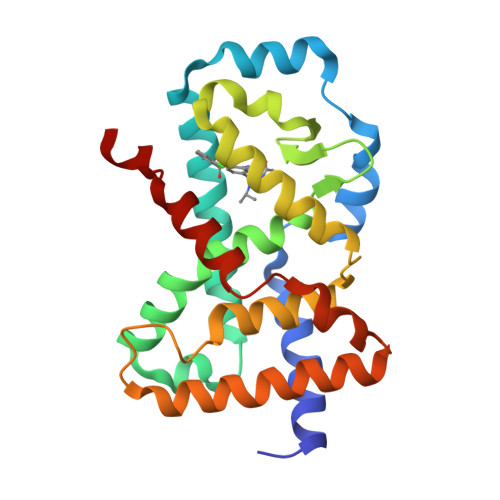
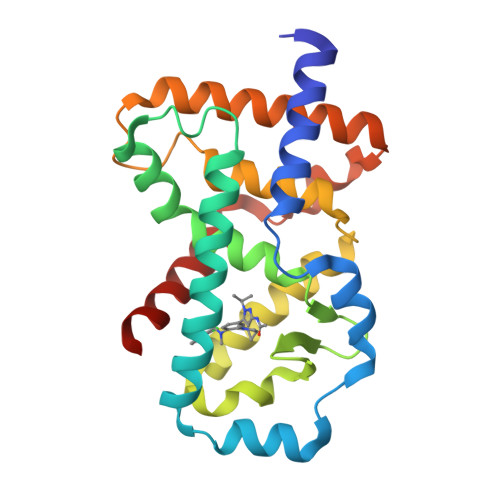
Discovery of novel pyrazole-containing benzamides as potent ROR gamma inverse agonists.
Wang, T., Banerjee, D., Bohnert, T., Chao, J., Enyedy, I., Fontenot, J., Guertin, K., Jones, H., Lin, E.Y., Marcotte, D., Talreja, T., Van Vloten, K.(2015) Bioorg Med Chem Lett 25: 2985-2990
- PubMed: 26048789
- DOI: https://doi.org/10.1016/j.bmcl.2015.05.028
- Primary Citation of Related Structures:
4ZOM - PubMed Abstract:
The nuclear receptor RORγ plays a central role in controlling a pro-inflammatory gene expression program in several lymphocyte lineages including TH17 cells. RORγ-dependent inflammation has been implicated in the pathogenesis of several major autoimmune diseases and thus RORγ is an attractive target for therapeutic intervention in these diseases. Starting from a lead biaryl compound 4a, replacement of the head phenyl moiety with a substituted aminopyrazole group resulted in a series with improved physical properties. Further SAR exploration led to analogues (e.g., 4j and 5m) as potent RORγ inverse agonists.
Organizational Affiliation:
Chemistry and Molecular Therapeutics, Biogen, 12 Cambridge Center, Cambridge, MA 02142, USA. Electronic address: taowang2@gmail.com.