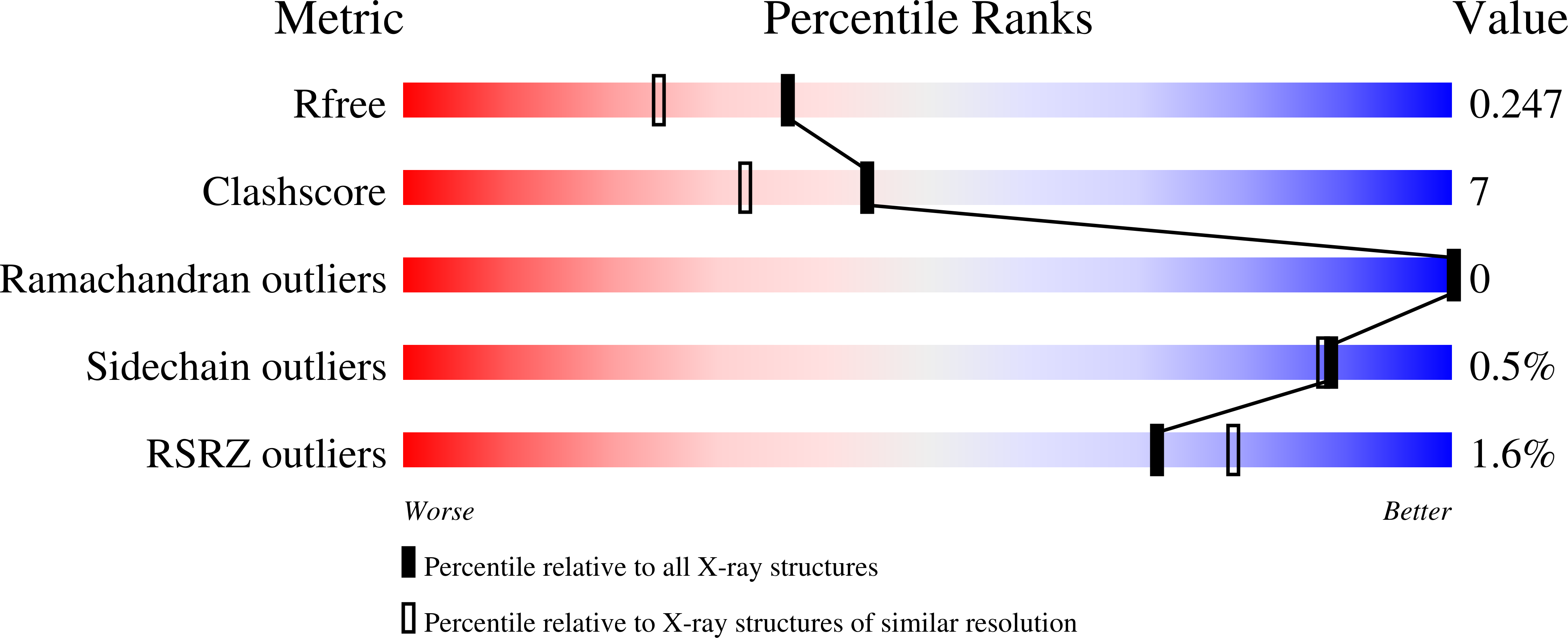
Gsk6853, a Chemical Probe for Inhibition of the Brpf1 Bromodomain.
Bamborough, P., Barnett, H.A., Becher, I., Bird, M.J., Chung, C., Craggs, P.D., Demont, E.H., Diallo, H., Fallon, D.J., Gordon, L.J., Grandi, P., Hobbs, C.I., Hooper-Greenhill, E., Jones, E.J., Law, R.P., Le Gall, A., Lugo, D., Michon, A., Mitchell, D.J., Prinjha, R.K., Sheppard, R.J., Watson, A.J.B., Watson, R.J.(2016) ACS Med Chem Lett 7: 552
- PubMed: 27326325
- DOI: https://doi.org/10.1021/acsmedchemlett.6b00092
- Primary Citation of Related Structures:
5G4R, 5G4S - PubMed Abstract:
The BRPF (Bromodomain and PHD Finger-containing) protein family are important scaffolding proteins for assembly of MYST histone acetyltransferase complexes. A selective benzimidazolone BRPF1 inhibitor showing micromolar activity in a cellular target engagement assay was recently described. Herein, we report the optimization of this series leading to the identification of a superior BRPF1 inhibitor suitable for in vivo studies.
Organizational Affiliation:
Epinova Discovery Performance Unit, Quantitative Pharmacology, Experimental Medicine Unit, Flexible Discovery Unit, and Platform Technology and Science, GlaxoSmithKline , Gunnels Wood Road, Stevenage, Hertfordshire SG1 2NY, U.K.