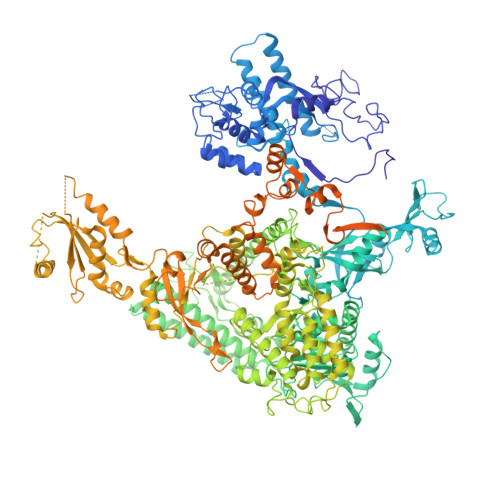
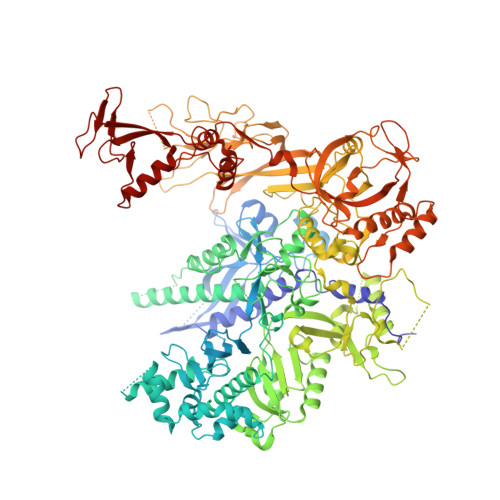
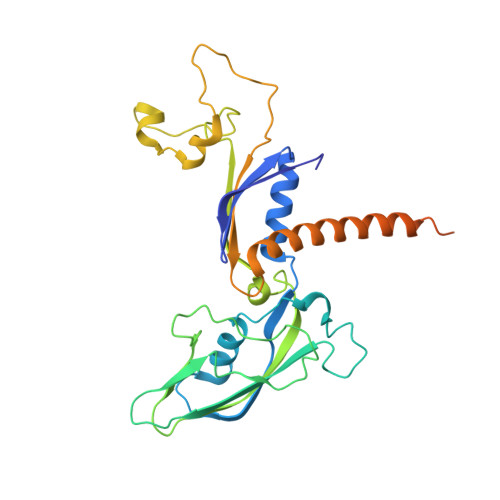
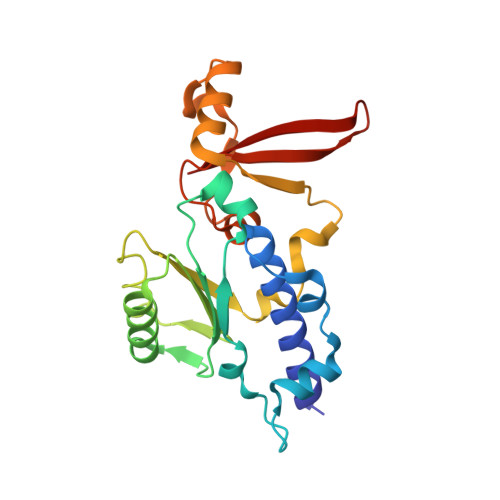
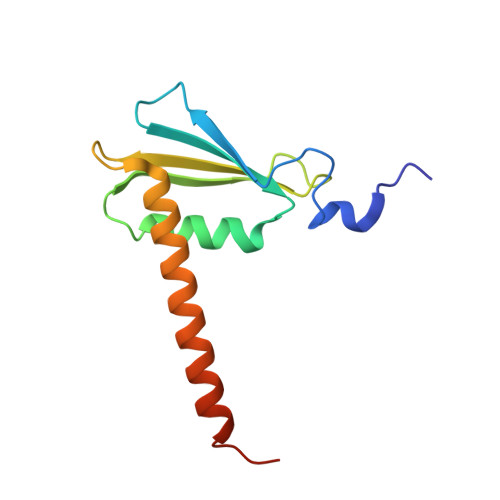
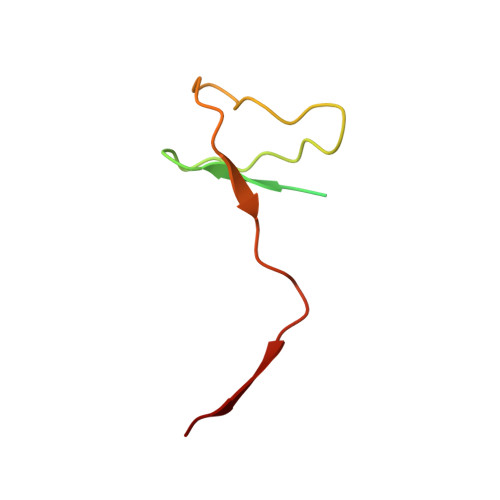
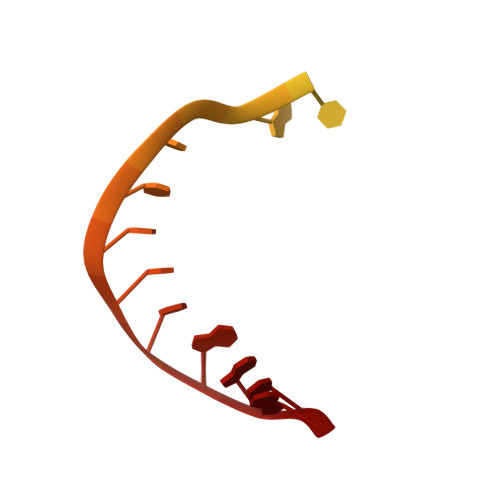
Structural basis of transcriptional stalling and bypass of abasic DNA lesion by RNA polymerase II.
Wang, W., Walmacq, C., Chong, J., Kashlev, M., Wang, D.(2018) Proc Natl Acad Sci U S A 115: E2538-E2545
- PubMed: 29487211
- DOI: https://doi.org/10.1073/pnas.1722050115
- Primary Citation of Related Structures:
6BLO, 6BLP, 6BM2, 6BM4, 6BQF - PubMed Abstract:
Abasic sites are among the most abundant DNA lesions and interfere with DNA replication and transcription, but the mechanism of their action on transcription remains unknown. Here we applied a combined structural and biochemical approach for a comprehensive investigation of how RNA polymerase II (Pol II) processes an abasic site, leading to slow bypass of lesion. Encounter of Pol II with an abasic site involves two consecutive slow steps: insertion of adenine opposite a noninstructive abasic site (the A-rule), followed by extension of the 3'-rAMP with the next cognate nucleotide. Further studies provided structural insights into the A-rule: ATP is slowly incorporated into RNA in the absence of template guidance. Our structure revealed that ATP is bound to the Pol II active site, whereas the abasic site is located at an intermediate state above the Bridge Helix, a conserved structural motif that is cirtical for Pol II activity. The next extension step occurs in a template-dependent manner where a cognate substrate is incorporated, despite at a much slower rate compared with nondamaged template. During the extension step, neither the cognate substrate nor the template base is located at the canonical position, providing a structural explanation as to why this step is as slow as the insertion step. Taken together, our studies provide a comprehensive understanding of Pol II stalling and bypass of the abasic site in the DNA template.
Organizational Affiliation:
Division of Pharmaceutical Sciences, Skaggs School of Pharmacy and Pharmaceutical Sciences, University of California, San Diego, La Jolla, CA 92093.