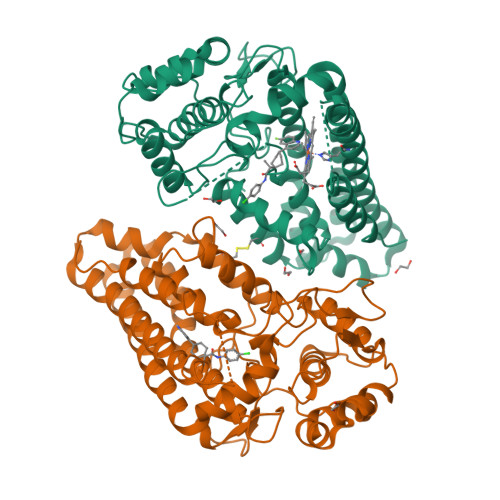
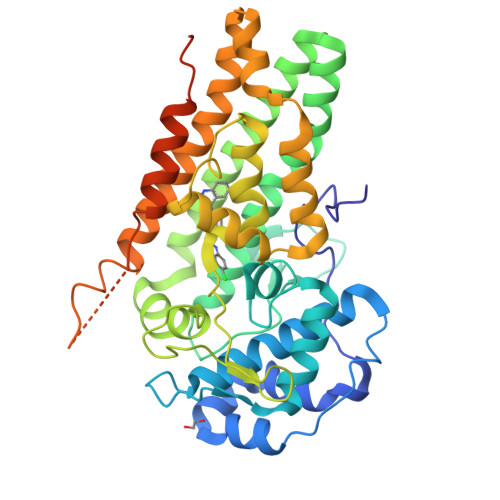
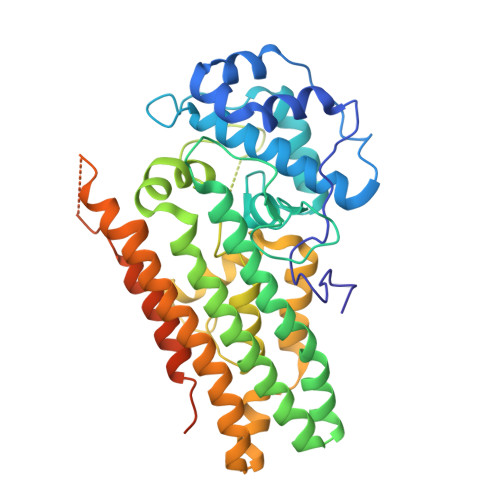
Mapping the Binding Trajectory of a Suicide Inhibitor in Human Indoleamine 2,3-Dioxygenase 1.
Pham, K.N., Yeh, S.R.(2018) J Am Chem Soc 140: 14538-14541
- PubMed: 30347977
- DOI: https://doi.org/10.1021/jacs.8b07994
- Primary Citation of Related Structures:
6DPQ, 6DPR, 6MQ6 - PubMed Abstract:
Human indoleamine 2,3-dioxygenase 1 (hIDO1) is an important heme-containing enzyme that is a key drug target for cancer immunotherapy. Several hIDO1 inhibitors have entered clinical trials, among which BMS-986205 (BMS) stands out as the only suicide inhibitor. Despite its "best-in-class" activity, the action mechanism of BMS remains elusive. Here, we report three crystal structures of hIDO1-BMS complexes that define the complete binding trajectory of the inhibitor. BMS first binds in a solvent exposed surface cleft near the active site in an extended conformation. The initial binding partially unfolds the active site, which triggers heme release, thereby exposing a new binding pocket. The inhibitor then undergoes a large scale movement to this new binding pocket, where it binds by adopting a high energy kinked conformation. Finally, the inhibitor relaxes to a bent conformation, via an additional large scale rearrangement, culminating in the energy minimum state. The structural data offer a molecular explanation for the remarkable efficacy and suicide inhibition activity of the inhibitor. They also suggest a novel strategy that can be applied for drug development targeting hIDO1 and related enzymes.
Organizational Affiliation:
Department of Physiology and Biophysics , Albert Einstein College of Medicine , Bronx , New York 10461 , United States.