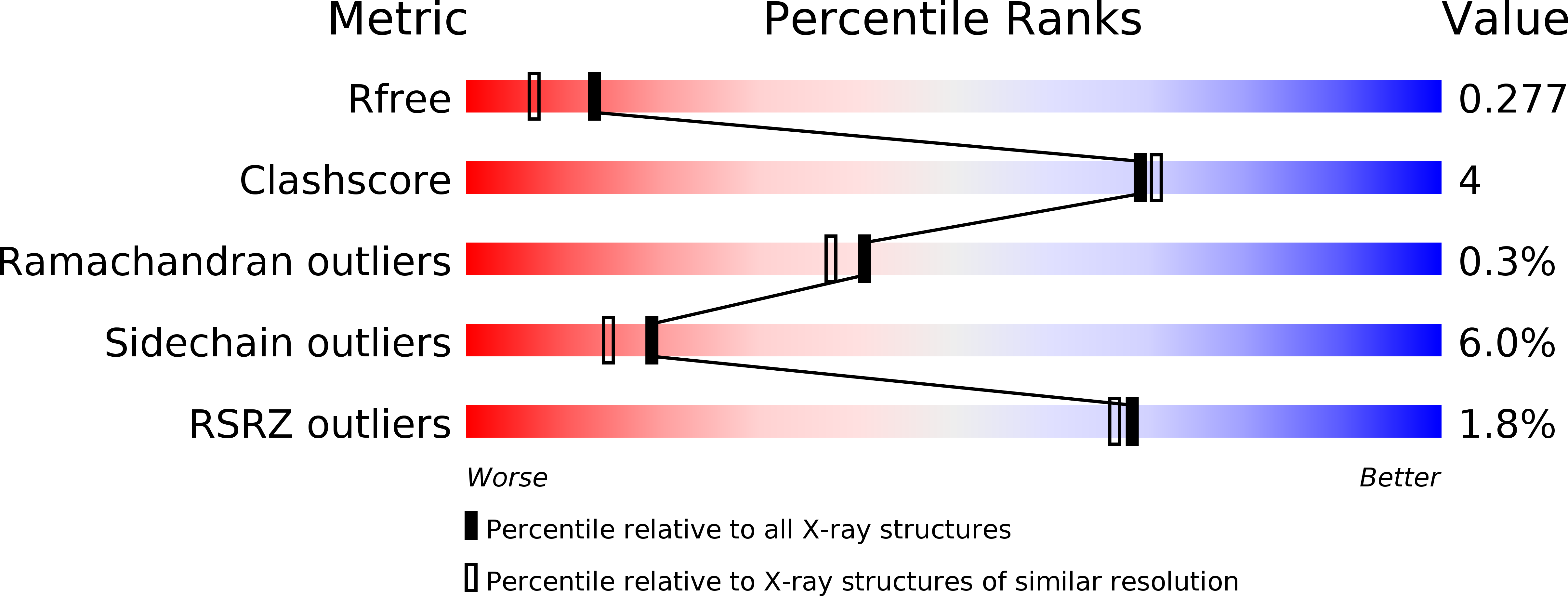
A suicide enzyme catalyzes multiple reactions for biotin biosynthesis in cyanobacteria.
Sakaki, K., Ohishi, K., Shimizu, T., Kobayashi, I., Mori, N., Matsuda, K., Tomita, T., Watanabe, H., Tanaka, K., Kuzuyama, T., Nishiyama, M.(2020) Nat Chem Biol 16: 415-422
- PubMed: 32042199
- DOI: https://doi.org/10.1038/s41589-019-0461-9
- Primary Citation of Related Structures:
6IR4, 6ITD, 6K36, 6K37, 6K38 - PubMed Abstract:
In biotin biosynthesis, the conversion of pimeloyl intermediates to biotin is catalyzed by a universal set of four enzymes: BioF, BioA, BioD and BioB. We found that the gene homologous to bioA, the product of which is involved in the conversion of 8-amino-7-oxononanoate (AON) to 7,8-diaminononanoate (DAN), is missing in the genome of the cyanobacterium Synechocystis sp. PCC 6803. We provide structural and biochemical evidence showing that a novel dehydrogenase, BioU, is involved in biotin biosynthesis and functionally replaces BioA. This enzyme catalyzes three reactions: formation of covalent linkage with AON to yield a BioU-DAN conjugate at the ε-amino group of Lys124 of BioU using NAD(P)H, carboxylation of the conjugate to form BioU-DAN-carbamic acid, and release of DAN-carbamic acid using NAD(P) + . In this biosynthetic pathway, BioU is a suicide enzyme that loses the Lys124 amino group after a single round of reaction.
Organizational Affiliation:
Biotechnology Research Center, The University of Tokyo, Bunkyo-ku, Tokyo, Japan.