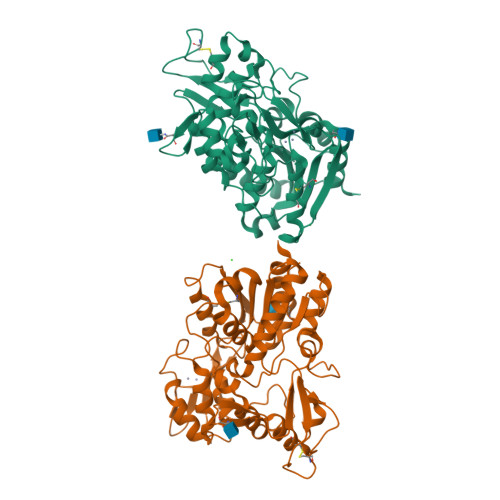
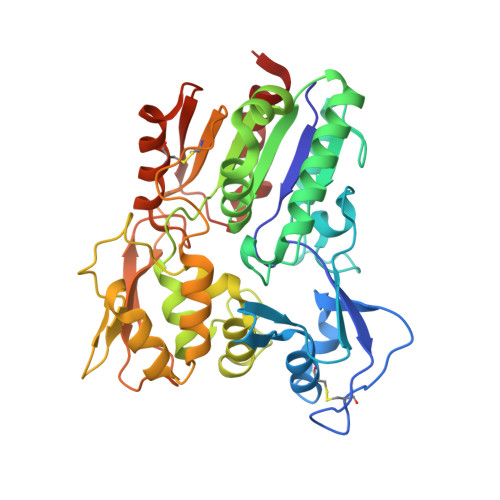
Structural Basis of Lysosomal Phospholipase A2Inhibition by Zn2.
Bouley, R.A., Hinkovska-Galcheva, V., Shayman, J.A., Tesmer, J.J.G.(2019) Biochemistry 58: 1709-1717
- PubMed: 30830753
- DOI: https://doi.org/10.1021/acs.biochem.8b01124
- Primary Citation of Related Structures:
6MTW - PubMed Abstract:
Lysosomal phospholipase A 2 (LPLA 2 /PLA2G15) is a key enzyme involved in lipid homeostasis and is characterized by both phospholipase A2 and transacylase activity and by an acidic pH optimum. Divalent cations such as Ca 2+ and Mg 2+ have previously been shown to have little effect on the activity of LPLA 2 , but the discovery of a novel crystal form of LPLA 2 with Zn 2+ bound in the active site suggested a role for this divalent cation in regulating enzyme activity. In this complex, the cation directly coordinates the serine and histidine of the α/β-hydrolase triad and stabilizes a closed conformation. This closed conformation is characterized by an inward shift of the lid loop, which extends over the active site and effectively blocks access to one of its lipid acyl chain binding tracks. Therefore, we hypothesized that Zn 2+ would inhibit LPLA 2 activity at a neutral but not acidic pH because histidine would be positively charged at lower pH. Indeed, Zn 2+ was found to inhibit the esterase activity of LPLA 2 in a noncompetitive manner exclusively at a neutral pH (between 6.5 and 8.0). Because lysosomes are reservoirs of Zn 2+ in cells, the pH optimum of LPLA 2 might allow it to catalyze acyl transfer unimpeded within the organelle. We conjecture that Zn 2+ inhibition of LPLA 2 at higher pH maintains a lower activity of the esterase in environments where its activity is not typically required.
Organizational Affiliation:
Life Sciences Institute and Department of Pharmacology , University of Michigan , Ann Arbor , Michigan 48109 , United States.