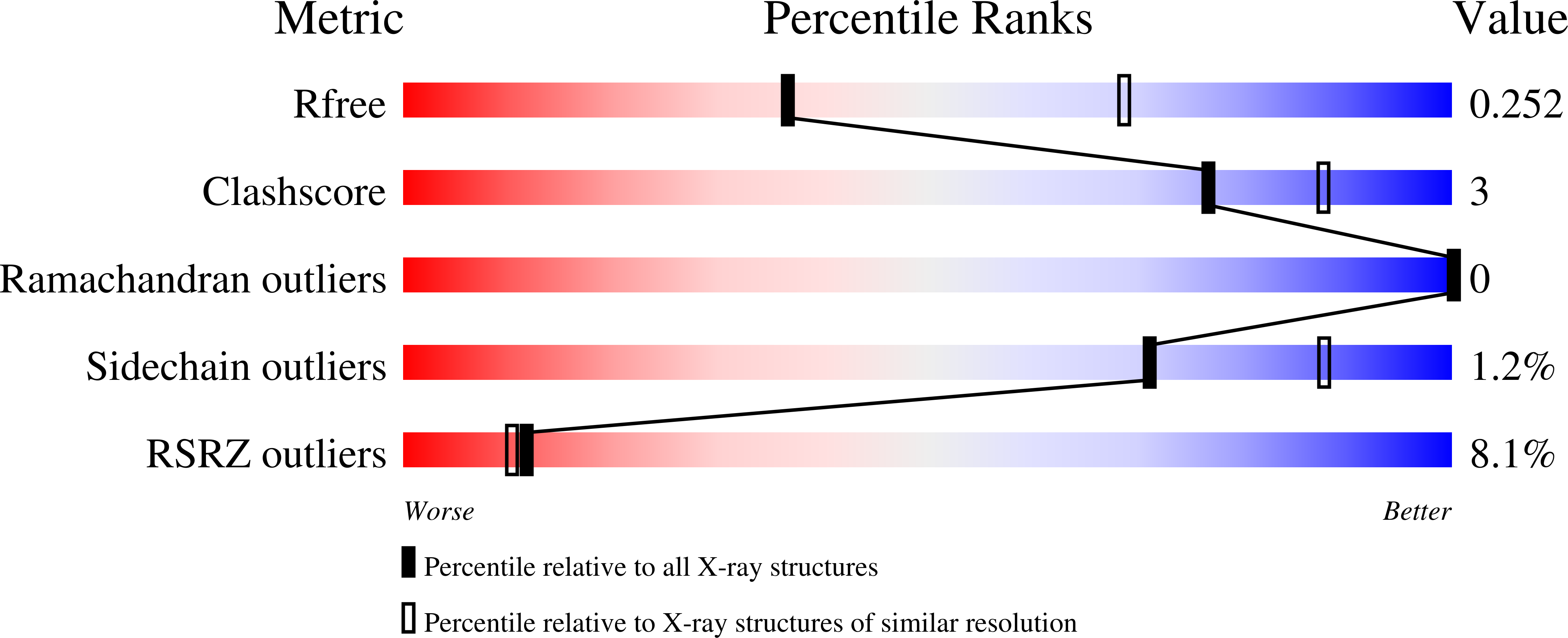
Structural investigation of 2-naphthyl phenyl ether inhibitors bound to WT and Y181C reverse transcriptase highlights key features of the NNRTI binding site.
Duong, V.N., Ippolito, J.A., Chan, A.H., Lee, W.G., Spasov, K.A., Jorgensen, W.L., Anderson, K.S.(2020) Protein Sci 29: 1902-1910
- PubMed: 32643196
- DOI: https://doi.org/10.1002/pro.3910
- Primary Citation of Related Structures:
6X47, 6X49, 6X4A, 6X4B, 6X4C, 6X4D, 6X4E, 6X4F - PubMed Abstract:
Human immunodeficiency virus (HIV)-1 remains as a global health issue that is primarily treated with highly active antiretroviral therapy, a combination of drugs that target the viral life cycle. One class of these drugs are non-nucleoside reverse transcriptase inhibitors (NNRTIs) that target the viral reverse transcriptase (RT). First generation NNRTIs were troubled with poor pharmacological properties and drug resistance, incentivizing the development of improved compounds. One class of developed compounds are the 2-naphthyl phenyl ethers, showing promising efficacy against the Y181C RT mutation. Further biochemical and structural work demonstrated differences in potency against the Y181C mutation and binding mode of the compounds. This work aims to understand the relationship between the binding mode and ability to overcome drug resistance using macromolecular x-ray crystallography. Comparison of 2-naphthyl phenyl ethers bound to Y181C RT reveal that compounds that interact with the invariant W229 are more capable of retaining efficacy against the resistance mutation. Additional modifications to these compounds at the 4-position, computationally designed to compensate for the Y181C mutation, do not demonstrate improved potency. Ultimately, we highlight important considerations for the development of future HIV-1 drugs that are able to combat drug resistance.
Organizational Affiliation:
Department of Pharmacology, Yale University School of Medicine, New Haven, Connecticut, USA.