Potent Tetrahydroquinolone Eliminates Apicomplexan Parasites.
McPhillie, M.J., Zhou, Y., Hickman, M.R., Gordon, J.A., Weber, C.R., Li, Q., Lee, P.J., Amporndanai, K., Johnson, R.M., Darby, H., Woods, S., Li, Z.H., Priestley, R.S., Ristroph, K.D., Biering, S.B., El Bissati, K., Hwang, S., Hakim, F.E., Dovgin, S.M., Lykins, J.D., Roberts, L., Hargrave, K., Cong, H., Sinai, A.P., Muench, S.P., Dubey, J.P., Prud'homme, R.K., Lorenzi, H.A., Biagini, G.A., Moreno, S.N., Roberts, C.W., Antonyuk, S.V., Fishwick, C.W.G., McLeod, R.(2020) Front Cell Infect Microbiol 10: 203-203
- PubMed: 32626661
- DOI: https://doi.org/10.3389/fcimb.2020.00203
- Primary Citation of Related Structures:
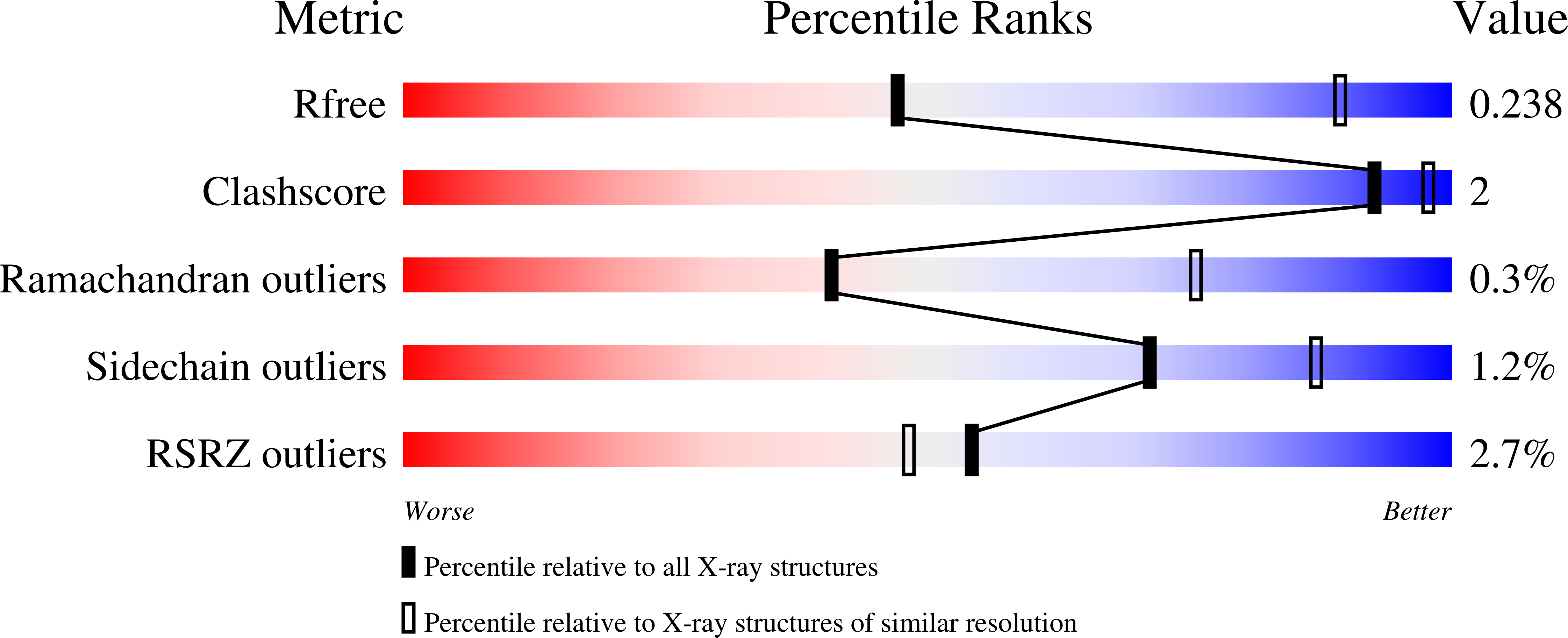
6XVF - PubMed Abstract:
Apicomplexan infections cause substantial morbidity and mortality, worldwide. New, improved therapies are needed. Herein, we create a next generation anti-apicomplexan lead compound, JAG21, a tetrahydroquinolone, with increased sp3-character to improve parasite selectivity. Relative to other cytochrome b inhibitors, JAG21 has improved solubility and ADMET properties, without need for pro-drug. JAG21 significantly reduces Toxoplasma gondii tachyzoites and encysted bradyzoites in vitro , and in primary and established chronic murine infections. Moreover, JAG21 treatment leads to 100% survival. Further, JAG21 is efficacious against drug-resistant Plasmodium falciparum in vitro . Causal prophylaxis and radical cure are achieved after P. berghei sporozoite infection with oral administration of a single dose (2.5 mg/kg) or 3 days treatment at reduced dose (0.625 mg/kg/day), eliminating parasitemia, and leading to 100% survival. Enzymatic, binding, and co-crystallography/pharmacophore studies demonstrate selectivity for apicomplexan relative to mammalian enzymes. JAG21 has significant promise as a pre-clinical candidate for prevention, treatment, and cure of toxoplasmosis and malaria.
Organizational Affiliation:
School of Chemistry, The University of Leeds, Leeds, United Kingdom.