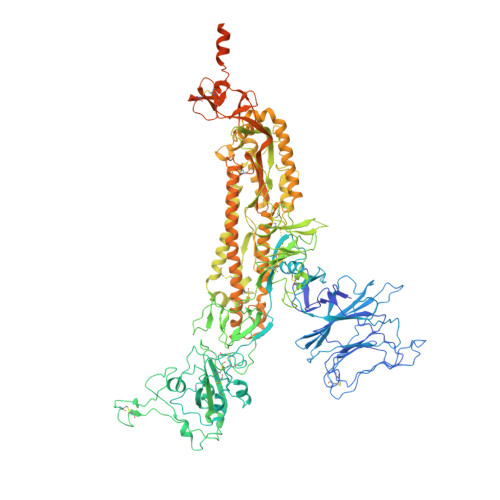
BA.2.12.1, BA.4 and BA.5 escape antibodies elicited by Omicron infection.
Cao, Y., Yisimayi, A., Jian, F., Song, W., Xiao, T., Wang, L., Du, S., Wang, J., Li, Q., Chen, X., Yu, Y., Wang, P., Zhang, Z., Liu, P., An, R., Hao, X., Wang, Y., Wang, J., Feng, R., Sun, H., Zhao, L., Zhang, W., Zhao, D., Zheng, J., Yu, L., Li, C., Zhang, N., Wang, R., Niu, X., Yang, S., Song, X., Chai, Y., Hu, Y., Shi, Y., Zheng, L., Li, Z., Gu, Q., Shao, F., Huang, W., Jin, R., Shen, Z., Wang, Y., Wang, X., Xiao, J., Xie, X.S.(2022) Nature 608: 593-602
- PubMed: 35714668
- DOI: https://doi.org/10.1038/s41586-022-04980-y
- Primary Citation of Related Structures:
7WR8, 7WRL, 7WRO, 7WRZ, 7X6A, 7XIW, 7XIX, 7XIY, 7XIZ, 7XNQ, 7XNR, 7XNS - PubMed Abstract:
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Omicron sublineages BA.2.12.1, BA.4 and BA.5 exhibit higher transmissibility than the BA.2 lineage 1 . The receptor binding and immune-evasion capability of these recently emerged variants require immediate investigation. Here, coupled with structural comparisons of the spike proteins, we show that BA.2.12.1, BA.4 and BA.5 (BA.4 and BA.5 are hereafter referred collectively to as BA.4/BA.5) exhibit similar binding affinities to BA.2 for the angiotensin-converting enzyme 2 (ACE2) receptor. Of note, BA.2.12.1 and BA.4/BA.5 display increased evasion of neutralizing antibodies compared with BA.2 against plasma from triple-vaccinated individuals or from individuals who developed a BA.1 infection after vaccination. To delineate the underlying antibody-evasion mechanism, we determined the escape mutation profiles 2 , epitope distribution 3 and Omicron-neutralization efficiency of 1,640 neutralizing antibodies directed against the receptor-binding domain of the viral spike protein, including 614 antibodies isolated from people who had recovered from BA.1 infection. BA.1 infection after vaccination predominantly recalls humoral immune memory directed against ancestral (hereafter referred to as wild-type (WT)) SARS-CoV-2 spike protein. The resulting elicited antibodies could neutralize both WT SARS-CoV-2 and BA.1 and are enriched on epitopes on spike that do not bind ACE2. However, most of these cross-reactive neutralizing antibodies are evaded by spike mutants L452Q, L452R and F486V. BA.1 infection can also induce new clones of BA.1-specific antibodies that potently neutralize BA.1. Nevertheless, these neutralizing antibodies are largely evaded by BA.2 and BA.4/BA.5 owing to D405N and F486V mutations, and react weakly to pre-Omicron variants, exhibiting narrow neutralization breadths. The therapeutic neutralizing antibodies bebtelovimab 4 and cilgavimab 5 can effectively neutralize BA.2.12.1 and BA.4/BA.5, whereas the S371F, D405N and R408S mutations undermine most broadly sarbecovirus-neutralizing antibodies. Together, our results indicate that Omicron may evolve mutations to evade the humoral immunity elicited by BA.1 infection, suggesting that BA.1-derived vaccine boosters may not achieve broad-spectrum protection against new Omicron variants.
Organizational Affiliation:
Biomedical Pioneering Innovation Center (BIOPIC), Peking University, Beijing, P. R. China. yunlongcao@pku.edu.cn.