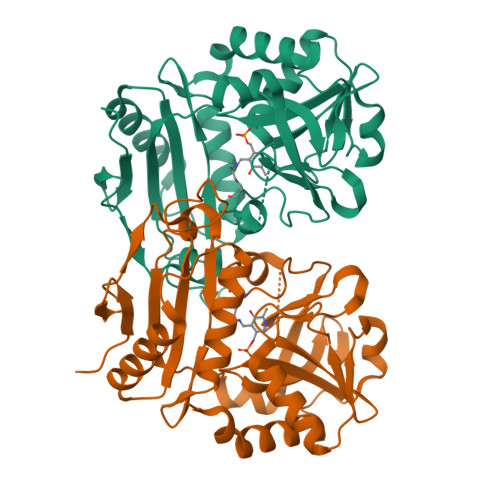
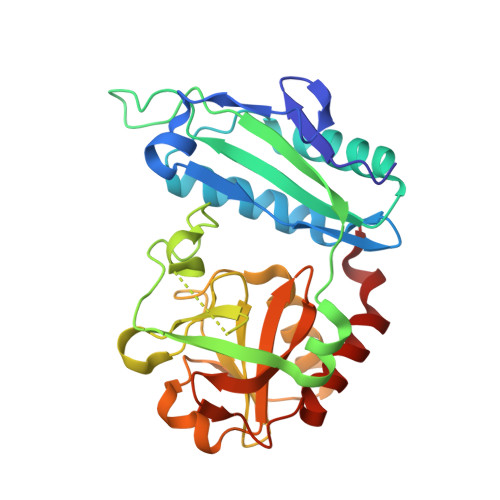
To the Understanding of Catalysis by D-Amino Acid Transaminases: A Case Study of the Enzyme from Aminobacterium colombiense.
Shilova, S.A., Khrenova, M.G., Matyuta, I.O., Nikolaeva, A.Y., Rakitina, T.V., Klyachko, N.L., Minyaev, M.E., Boyko, K.M., Popov, V.O., Bezsudnova, E.Y.(2023) Molecules 28
- PubMed: 36903355
- DOI: https://doi.org/10.3390/molecules28052109
- Primary Citation of Related Structures:
8AHR, 8AYK - PubMed Abstract:
Pyridoxal-5'-phosphate (PLP)-dependent transaminases are highly efficient biocatalysts for stereoselective amination. D-amino acid transaminases can catalyze stereoselective transamination producing optically pure D-amino acids. The knowledge of substrate binding mode and substrate differentiation mechanism in D-amino acid transaminases comes down to the analysis of the transaminase from Bacillus subtilis . However, at least two groups of D-amino acid transaminases differing in the active site organization are known today. Here, we present a detailed study of D-amino acid transaminase from the gram-negative bacterium Aminobacterium colombiense with a substrate binding mode different from that for the transaminase from B. subtilis . We study the enzyme using kinetic analysis, molecular modeling, and structural analysis of holoenzyme and its complex with D-glutamate. We compare the multipoint binding of D-glutamate with the binding of other substrates, D-aspartate and D-ornithine. QM/MM MD simulation reveals that the substrate can act as a base and its proton can be transferred from the amino group to the α-carboxylate group. This process occurs simultaneously with the nucleophilic attack of the PLP carbon atom by the nitrogen atom of the substrate forming gem-diamine at the transimination step. This explains the absence of the catalytic activity toward ( R )-amines that lack an α-carboxylate group. The obtained results clarify another substrate binding mode in D-amino acid transaminases and underpinned the substrate activation mechanism.
Organizational Affiliation:
Bach Institute of Biochemistry, Research Centre of Biotechnology of the Russian Academy of Sciences, 119071 Moscow, Russia.