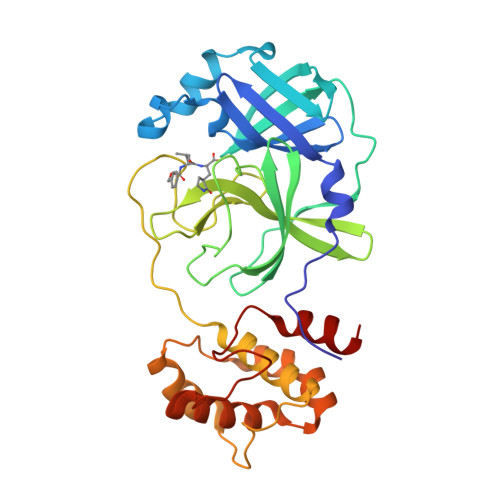
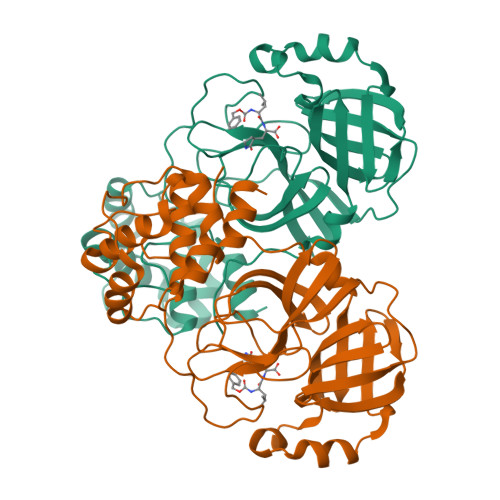
Contribution of the catalytic dyad of SARS-CoV-2 main protease to binding covalent and noncovalent inhibitors.
Kovalevsky, A., Aniana, A., Coates, L., Bonnesen, P.V., Nashed, N.T., Louis, J.M.(2023) J Biological Chem 299: 104886-104886
- PubMed: 37271339
- DOI: https://doi.org/10.1016/j.jbc.2023.104886
- Primary Citation of Related Structures:
8GFK, 8GFN, 8GFO, 8GFR, 8GFU - PubMed Abstract:
The effect of mutations of the catalytic dyad residues of SARS-CoV-2 main protease (MPro WT ) on the thermodynamics of binding of covalent inhibitors comprising nitrile [nirmatrelvir (NMV), NBH2], aldehyde (GC373), and ketone (BBH1) warheads to MPro is examined together with room temperature X-ray crystallography. When lacking the nucleophilic C145, NMV binding is ∼400-fold weaker corresponding to 3.5 kcal/mol and 13.3 °C decrease in free energy (ΔG) and thermal stability (T m ), respectively, relative to MPro WT . The H41A mutation results in a 20-fold increase in the dissociation constant (K d ), and 1.7 kcal/mol and 1.4 °C decreases in ΔG and T m , respectively. Increasing the pH from 7.2 to 8.2 enhances NMV binding to MPro H41A , whereas no significant change is observed in binding to MPro WT . Structures of the four inhibitor complexes with MPro 1-304/C145A show that the active site geometries of the complexes are nearly identical to that of MPro WT with the nucleophilic sulfur of C145 positioned to react with the nitrile or the carbonyl carbon. These results support a two-step mechanism for the formation of the covalent complex involving an initial non-covalent binding followed by a nucleophilic attack by the thiolate anion of C145 on the warhead carbon. Noncovalent inhibitor ensitrelvir (ESV) exhibits a binding affinity to MPro WT that is similar to NMV but differs in its thermodynamic signature from NMV. The binding of ESV to MPro C145A also results in a significant, but smaller, increase in K d and decrease in ΔG and T m , relative to NMV.
Organizational Affiliation:
Neutron Scattering Division, Oak Ridge National Laboratory, Oak Ridge, Tennessee, USA. Electronic address: kovalevskyay@ornl.gov.