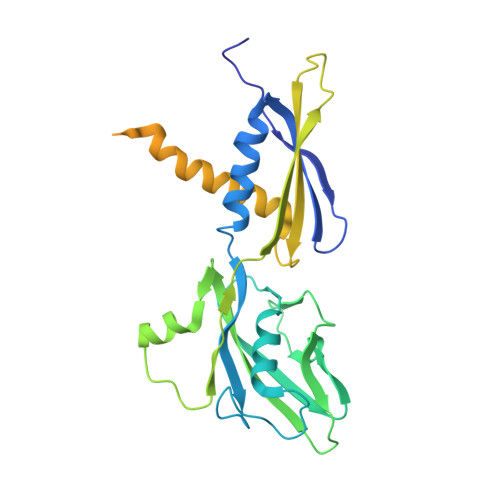
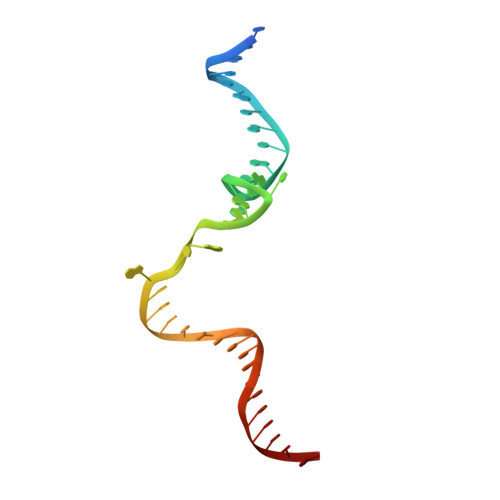
Concerted transformation of a hyper-paused transcription complex and its reinforcing protein.
Zuber, P.K., Said, N., Hilal, T., Wang, B., Loll, B., Gonzalez-Higueras, J., Ramirez-Sarmiento, C.A., Belogurov, G.A., Artsimovitch, I., Wahl, M.C., Knauer, S.H.(2024) Nat Commun 15: 3040-3040
- PubMed: 38589445
- DOI: https://doi.org/10.1038/s41467-024-47368-4
- Primary Citation of Related Structures:
8PDY, 8PEN, 8PFG, 8PFJ, 8PH9, 8PHK, 8PIB, 8PID, 8PIL, 8PIM - PubMed Abstract:
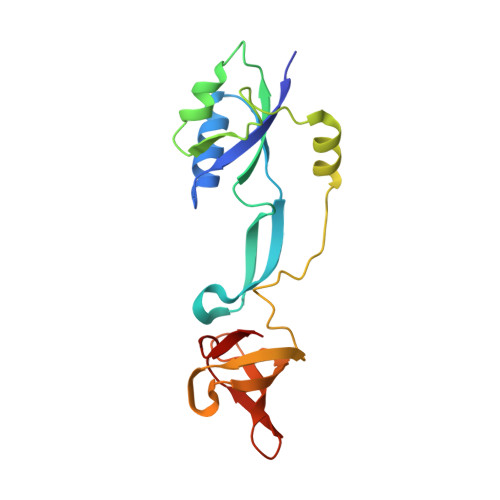
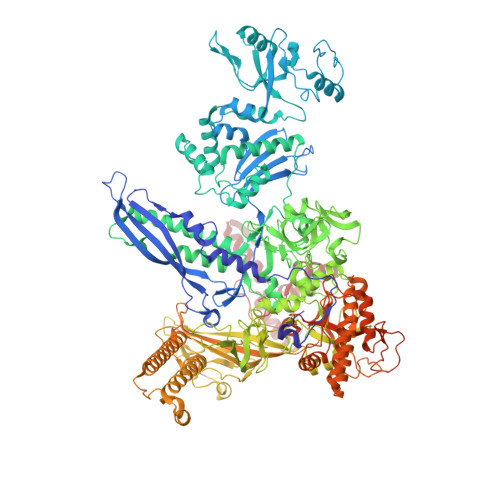
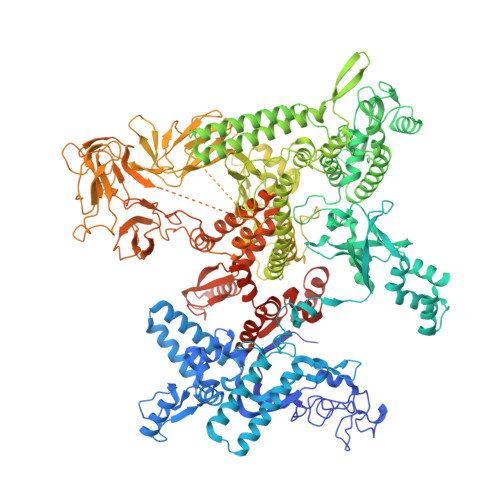
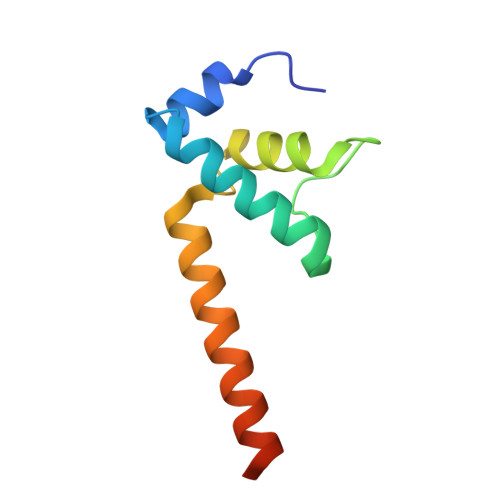
RfaH, a paralog of the universally conserved NusG, binds to RNA polymerases (RNAP) and ribosomes to activate expression of virulence genes. In free, autoinhibited RfaH, an α-helical KOW domain sequesters the RNAP-binding site. Upon recruitment to RNAP paused at an ops site, KOW is released and refolds into a β-barrel, which binds the ribosome. Here, we report structures of ops-paused transcription elongation complexes alone and bound to the autoinhibited and activated RfaH, which reveal swiveled, pre-translocated pause states stabilized by an ops hairpin in the non-template DNA. Autoinhibited RfaH binds and twists the ops hairpin, expanding the RNA:DNA hybrid to 11 base pairs and triggering the KOW release. Once activated, RfaH hyper-stabilizes the pause, which thus requires anti-backtracking factors for escape. Our results suggest that the entire RfaH cycle is solely determined by the ops and RfaH sequences and provide insights into mechanisms of recruitment and metamorphosis of NusG homologs across all life.
Organizational Affiliation:
Biochemistry IV-Biophysical Chemistry, Universität Bayreuth, Bayreuth, Germany.