Crystal Structure and Computational Analyses Provide Insights into the Catalytic Mechanism of 2,4-Diacetylphloroglucinol Hydrolase PhlG from Pseudomonas fluorescens.
He, Y.X., Huang, L., Xue, Y., Fei, X., Teng, Y.B., Rubin-Pitel, S.B., Zhao, H., Zhou, C.Z.(2010) J Biological Chem 285: 4603-4611
- PubMed: 20018877
- DOI: https://doi.org/10.1074/jbc.M109.044180
- Primary Citation of Related Structures:
3HWP - PubMed Abstract:
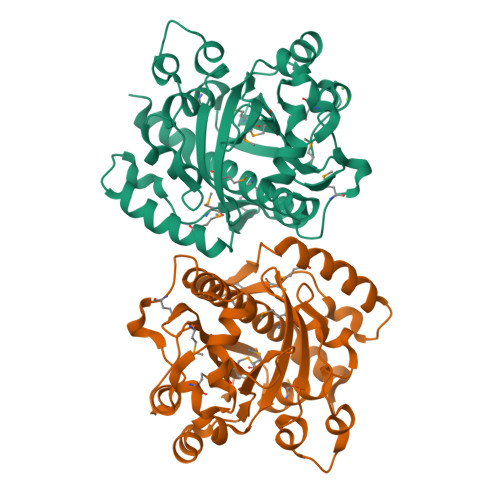
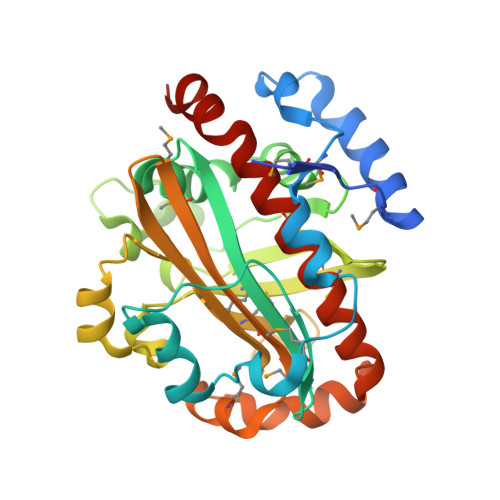
2,4-Diacetylphloroglucinol hydrolase PhlG from Pseudomonas fluorescens catalyzes hydrolytic carbon-carbon (C-C) bond cleavage of the antibiotic 2,4-diacetylphloroglucinol to form monoacetylphloroglucinol, a rare class of reactions in chemistry and biochemistry. To investigate the catalytic mechanism of this enzyme, we determined the three-dimensional structure of PhlG at 2.0 A resolution using x-ray crystallography and MAD methods. The overall structure includes a small N-terminal domain mainly involved in dimerization and a C-terminal domain of Bet v1-like fold, which distinguishes PhlG from the classical alpha/beta-fold hydrolases. A dumbbell-shaped substrate access tunnel was identified to connect a narrow interior amphiphilic pocket to the exterior solvent. The tunnel is likely to undergo a significant conformational change upon substrate binding to the active site. Structural analysis coupled with computational docking studies, site-directed mutagenesis, and enzyme activity analysis revealed that cleavage of the 2,4-diacetylphloroglucinol C-C bond proceeds via nucleophilic attack by a water molecule, which is coordinated by a zinc ion. In addition, residues Tyr(121), Tyr(229), and Asn(132), which are predicted to be hydrogen-bonded to the hydroxyl groups and unhydrolyzed acetyl group, can finely tune and position the bound substrate in a reactive orientation. Taken together, these results revealed the active sites and zinc-dependent hydrolytic mechanism of PhlG and explained its substrate specificity as well.
Organizational Affiliation:
Hefei National Laboratory for Physical Sciences at Microscale and School of Life Sciences, University of Science and Technology of China, Hefei, Anhui 230026, China.