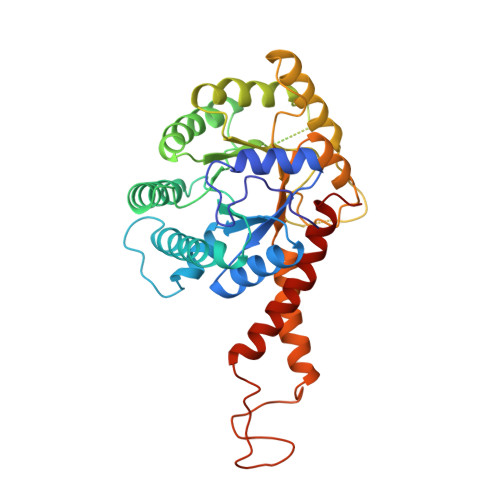
The crystal structure of a class II fructose-1,6-bisphosphate aldolase shows a novel binuclear metal-binding active site embedded in a familiar fold.
Cooper, S.J., Leonard, G.A., McSweeney, S.M., Thompson, A.W., Naismith, J.H., Qamar, S., Plater, A., Berry, A., Hunter, W.N.(1996) Structure 4: 1303-1315
- PubMed: 8939754
- DOI: https://doi.org/10.1016/s0969-2126(96)00138-4
- Primary Citation of Related Structures:
1ZEN - PubMed Abstract:
[corrected] Aldolases catalyze a variety of condensation and cleavage reactions, with exquisite control on the stereochemistry. These enzymes, therefore, are attractive catalysts for synthetic chemistry. There are two classes of aldolase: class I aldolases utilize Schiff base formation with an active-site lysine whilst class II enzymes require a divalent metal ion, in particular zinc. Fructose-1,6-bisphosphate aldolase (FBP-aldolase) is used in gluconeogenesis and glycolysis; the enzyme controls the condensation of dihydroxyacetone phosphate with glyceraldehyde-3-phosphate to yield fructose-1,6-bisphosphate. Structures are available for class I FBP-aldolases but there is a paucity of detail on the class II enzymes. Characterization is sought to enable a dissection of structure/activity relationships which may assist the construction of designed aldolases for use as biocatalysts in synthetic chemistry. The structure of the dimeric class II FBP-aldolase from Escherichia coli has been determined using data to 2.5 A resolution. The asymmetric unit is one subunit which presents a familiar fold, the (alpha/beta)8 barrel. The active centre, at the C-terminal end of the barrel, contains a novel bimetallic-binding site with two metal ions 6.2 A apart. One ion, the identity of which is not certain, is buried and may play a structural or activating role. The other metal ion is zinc and is positioned at the surface of the barrel to participate in catalysis. Comparison of the structure with a class II fuculose aldolase suggests that these enzymes may share a common mechanism. Nevertheless, the class II enzymes should be subdivided into two categories on consideration of subunit size and fold, quaternary structure and metal-ion binding sites.
Organizational Affiliation:
Department of Chemistry, University of Manchester, Oxford Road, Manchester, M13 9PL, UK. wnhunter@bad.dundee.ac.uk