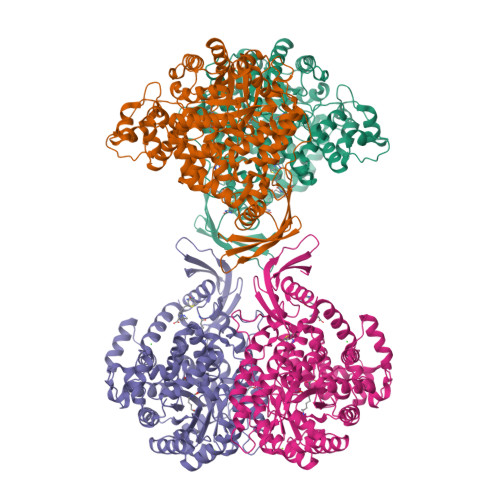
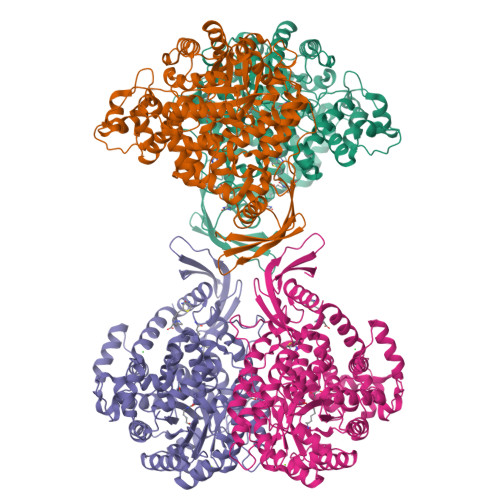
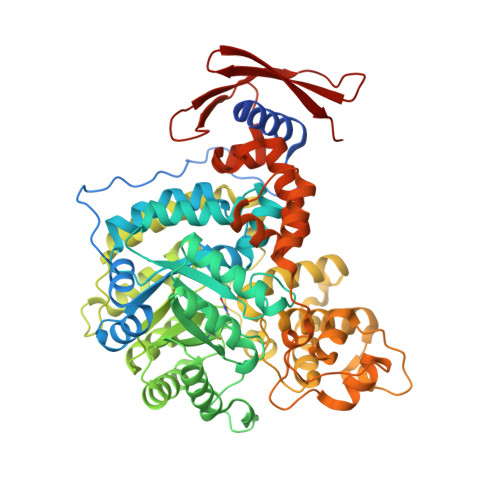
The role of biotin and oxamate in the carboxyltransferase reaction of pyruvate carboxylase.
Lietzan, A.D., Lin, Y., St.Maurice, M.(2014) Arch Biochem Biophys 562C: 70-79
- PubMed: 25157442
- DOI: https://doi.org/10.1016/j.abb.2014.08.008
- Primary Citation of Related Structures:
4LOC, 4M6V - PubMed Abstract:
Pyruvate carboxylase (PC) is a biotin-dependent enzyme that catalyzes the MgATP-dependent carboxylation of pyruvate to oxaloacetate, an important anaplerotic reaction in central metabolism. During catalysis, carboxybiotin is translocated to the carboxyltransferase domain where the carboxyl group is transferred to the acceptor substrate, pyruvate. Many studies on the carboxyltransferase domain of PC have demonstrated an enhanced oxaloacetate decarboxylation activity in the presence of oxamate and it has been shown that oxamate accepts a carboxyl group from carboxybiotin during oxaloacetate decarboxylation. The X-ray crystal structure of the carboxyltransferase domain from Rhizobium etli PC reveals that oxamate is positioned in the active site in an identical manner to the substrate, pyruvate, and kinetic data are consistent with the oxamate-stimulated decarboxylation of oxaloacetate proceeding through a simple ping-pong bi bi mechanism in the absence of the biotin carboxylase domain. Additionally, analysis of truncated PC enzymes indicates that the BCCP domain devoid of biotin does not contribute directly to the enzymatic reaction and conclusively demonstrates a biotin-independent oxaloacetate decarboxylation activity in PC. These findings advance the description of catalysis in PC and can be extended to the study of related biotin-dependent enzymes.
Organizational Affiliation:
Department of Biological Sciences, Marquette University, Milwaukee, WI 53201, USA.