The hydrogen peroxide hypersensitivity of OxyR2 in Vibrio vulnificus depends on conformational constraints
Jo, I., Kim, D., Bang, Y.-J., Ahn, J., Choi, S.H., Ha, N.-C.(2017) J Biological Chem 292: 7223-7232
- PubMed: 28264933
- DOI: https://doi.org/10.1074/jbc.M116.743765
- Primary Citation of Related Structures:
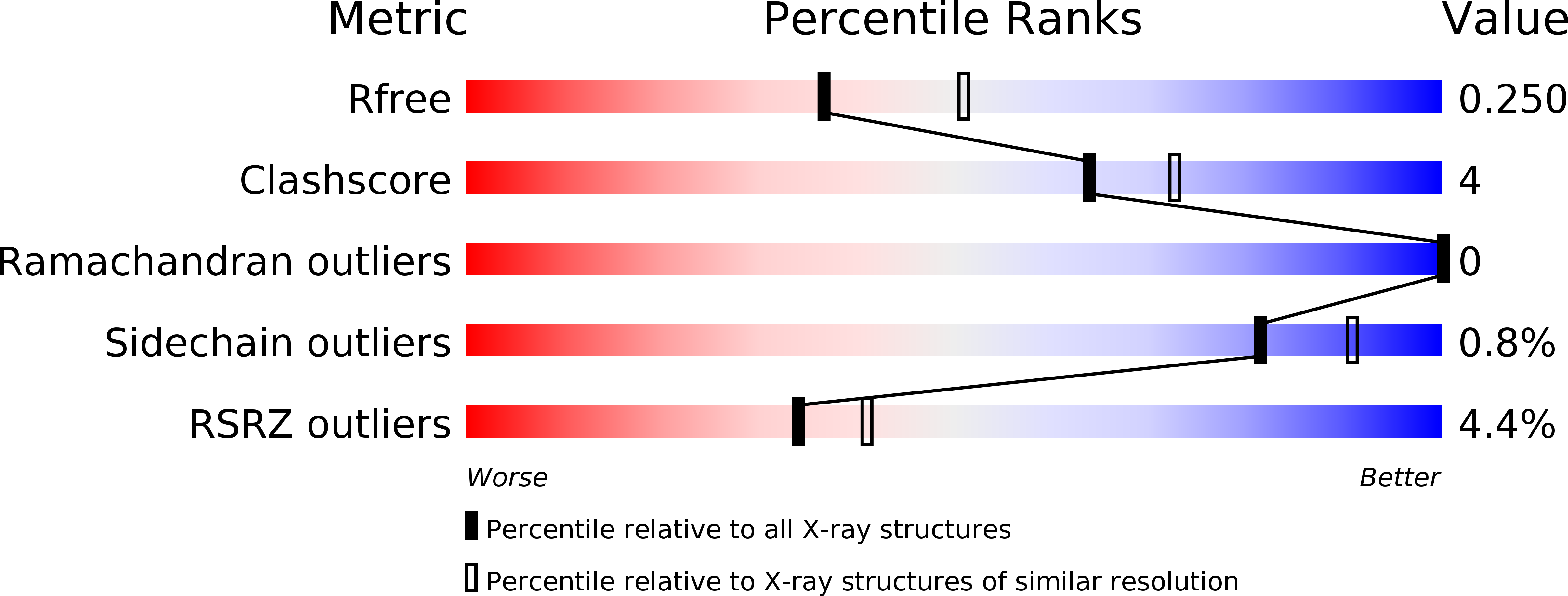
5B70, 5B7D, 5X0Q, 5X0V - PubMed Abstract:
Most Gram-negative bacteria respond to excessive levels of H 2 O 2 using the peroxide-sensing transcriptional regulator OxyR, which can induce the expression of antioxidant genes to restore normality. Vibrio vulnificus has two distinct OxyRs (OxyR1 and OxyR2), which are sensitive to different levels of H 2 O 2 and induce expression of two different peroxidases, Prx1 and Prx2. Although OxyR1 has both high sequence similarity and H 2 O 2 sensitivity comparable with that of other OxyR proteins, OxyR2 exhibits limited sequence similarity and is more sensitive to H 2 O 2 To investigate the basis for this difference, we determined crystal structures and carried out biochemical analyses of OxyR2. The determined structure of OxyR2 revealed a flipped conformation of the peptide bond before Glu-204, a position occupied by glycine in other OxyR proteins. Activity assays showed that the sensitivity to H 2 O 2 was reduced to the level of other OxyR proteins by the E204G mutation. We solved the structure of the OxyR2-E204G mutant with the same packing environment. The structure of the mutant revealed a dual conformation of the peptide bond before Gly-204, indicating the structural flexibility of the region. This structural duality extended to the backbone atoms of Gly-204 and the imidazole ring of His-205, which interact with H 2 O 2 and invariant water molecules near the peroxidatic cysteine, respectively. Structural comparison suggests that Glu-204 in OxyR2 provides rigidity to the region that is important in H 2 O 2 sensing, compared with the E204G structure or other OxyR proteins. Our findings provide a structural basis for the higher sensitivity of OxyR2 to H 2 O 2 and also suggest a molecular mechanism for bacterial regulation of expression of antioxidant genes at divergent concentrations of cellular H 2 O 2 .
Organizational Affiliation:
From the Department of Agricultural Biotechnology, Center for Food Safety and Toxicology, Research Institute for Agriculture and Life Sciences, and.