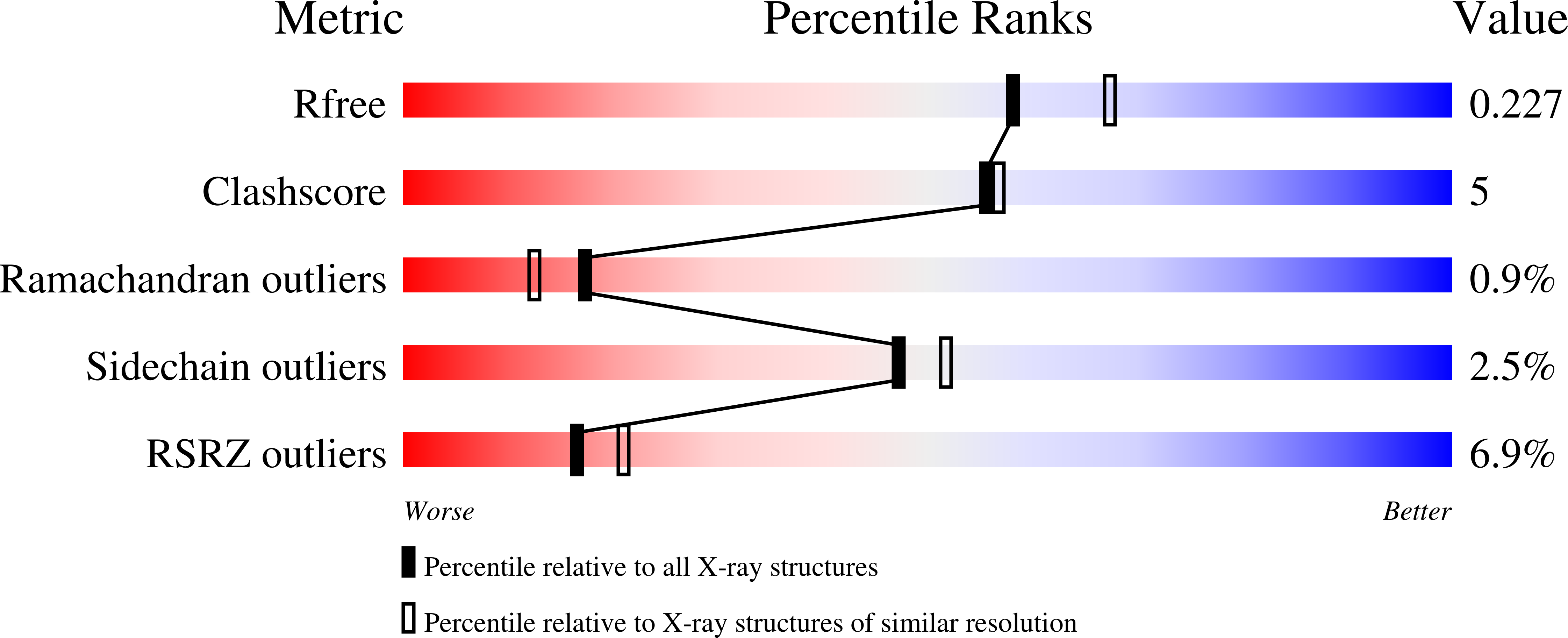
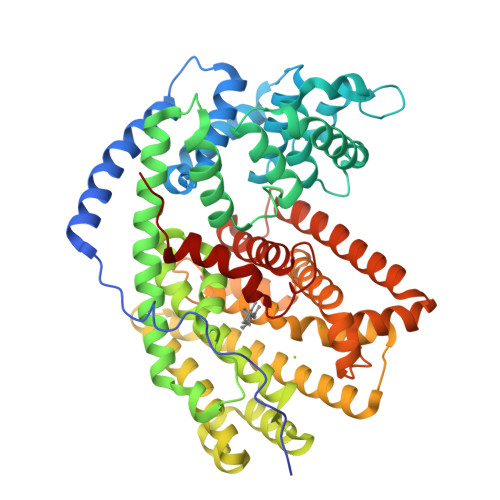
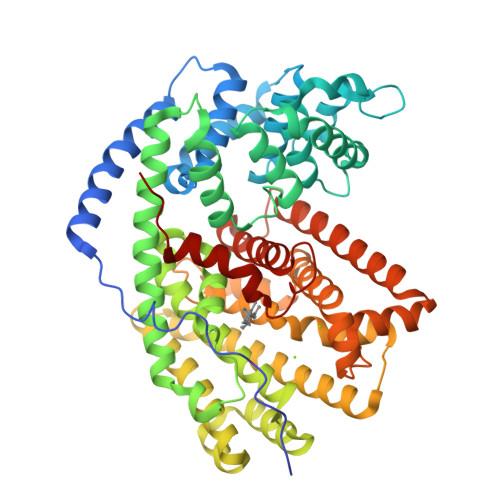
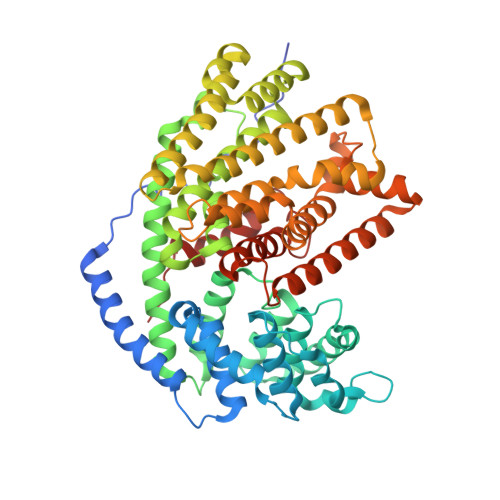
Biosynthetic potential of sesquiterpene synthases: product profiles of Egyptian Henbane premnaspirodiene synthase and related mutants.
Koo, H.J., Vickery, C.R., Xu, Y., Louie, G.V., O'Maille, P.E., Bowman, M., Nartey, C.M., Burkart, M.D., Noel, J.P.(2016) J Antibiot (Tokyo) 69: 524-533
- PubMed: 27328867
- DOI: https://doi.org/10.1038/ja.2016.68
- Primary Citation of Related Structures:
5IK0, 5IK6, 5IK9, 5IKA, 5IKH - PubMed Abstract:
The plant terpene synthase (TPS) family is responsible for the biosynthesis of a variety of terpenoid natural products possessing diverse biological functions. TPSs catalyze the ionization and, most commonly, rearrangement and cyclization of prenyl diphosphate substrates, forming linear and cyclic hydrocarbons. Moreover, a single TPS often produces several minor products in addition to a dominant product. We characterized the catalytic profiles of Hyoscyamus muticus premnaspirodiene synthase (HPS) and compared it with the profile of a closely related TPS, Nicotiana tabacum 5-epi-aristolochene synthase (TEAS). The profiles of two previously studied HPS and TEAS mutants, each containing nine interconverting mutations, dubbed HPS-M9 and TEAS-M9, were also characterized. All four TPSs were compared under varying temperature and pH conditions. In addition, we solved the X-ray crystal structures of TEAS and a TEAS quadruple mutant complexed with substrate and products to gain insight into the enzymatic features modulating product formation. These informative structures, along with product profiles, provide new insight into plant TPS catalytic promiscuity.
Organizational Affiliation:
Howard Hughes Medical Institute, The Salk Institute for Biological Studies, Jack H Skirball Center for Chemical Biology and Proteomics, La Jolla, CA, USA.