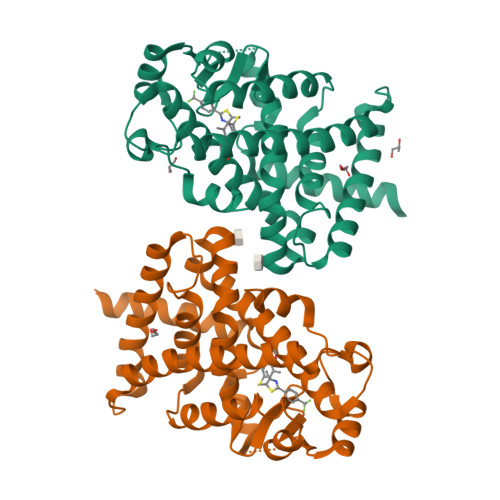
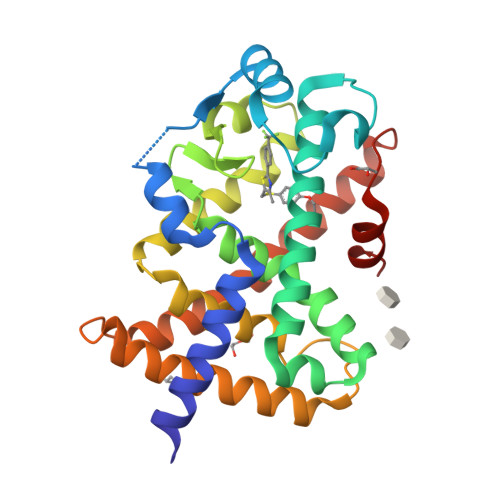
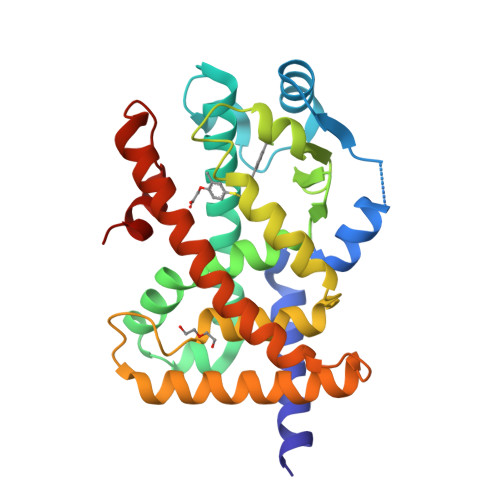
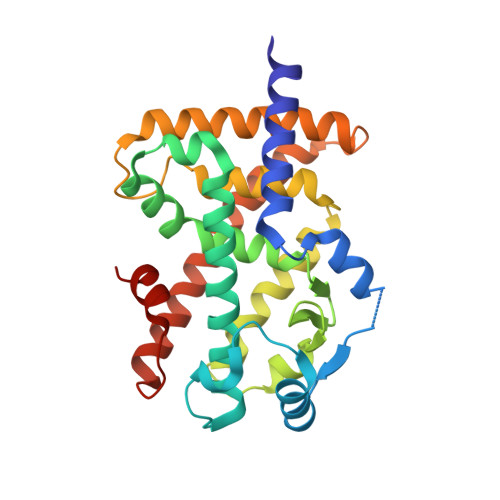
Structural basis for specific ligation of the peroxisome proliferator-activated receptor delta.
Wu, C.C., Baiga, T.J., Downes, M., La Clair, J.J., Atkins, A.R., Richard, S.B., Fan, W., Stockley-Noel, T.A., Bowman, M.E., Noel, J.P., Evans, R.M.(2017) Proc Natl Acad Sci U S A 114: E2563-E2570
- PubMed: 28320959
- DOI: https://doi.org/10.1073/pnas.1621513114
- Primary Citation of Related Structures:
5U3Q, 5U3R, 5U3S, 5U3T, 5U3U, 5U3V, 5U3W, 5U3X, 5U3Y, 5U3Z, 5U40, 5U41, 5U42, 5U43, 5U44, 5U45, 5U46 - PubMed Abstract:
The peroxisome proliferator-activated receptor (PPAR) family comprises three subtypes: PPARα, PPARγ, and PPARδ. PPARδ transcriptionally modulates lipid metabolism and the control of energy homeostasis; therefore, PPARδ agonists are promising agents for treating a variety of metabolic disorders. In the present study, we develop a panel of rationally designed PPARδ agonists. The modular motif affords efficient syntheses using building blocks optimized for interactions with subtype-specific residues in the PPARδ ligand-binding domain (LBD). A combination of atomic-resolution protein X-ray crystallographic structures, ligand-dependent LBD stabilization assays, and cell-based transactivation measurements delineate structure-activity relationships (SARs) for PPARδ-selective targeting and structural modulation. We identify key ligand-induced conformational transitions of a conserved tryptophan side chain in the LBD that trigger reorganization of the H2'-H3 surface segment of PPARδ. The subtype-specific conservation of H2'-H3 sequences suggests that this architectural remodeling constitutes a previously unrecognized conformational switch accompanying ligand-dependent PPARδ transcriptional regulation.
Organizational Affiliation:
Howard Hughes Medical Institute, The Salk Institute for Biological Studies, La Jolla, CA 92037.