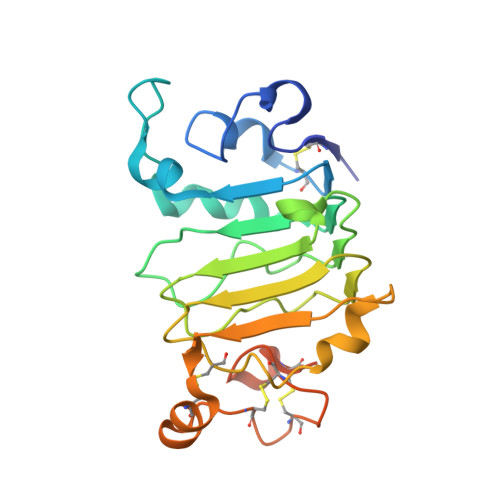
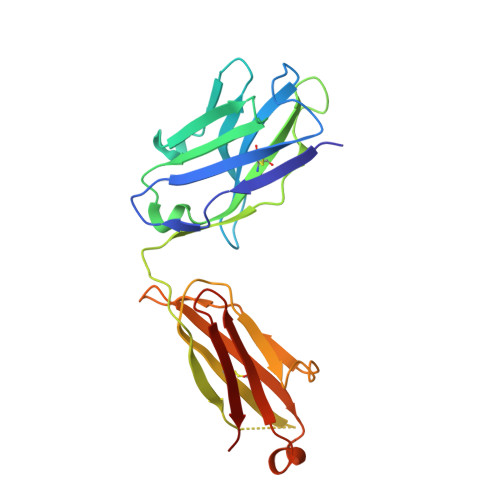
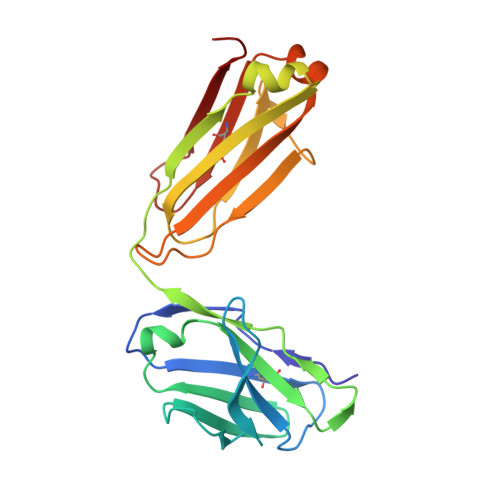
Molecular Basis for Necitumumab Inhibition of EGFR Variants Associated with Acquired Cetuximab Resistance.
Bagchi, A., Haidar, J.N., Eastman, S.W., Vieth, M., Topper, M., Iacolina, M.D., Walker, J.M., Forest, A., Shen, Y., Novosiadly, R.D., Ferguson, K.M.(2018) Mol Cancer Ther 17: 521-531
- PubMed: 29158469
- DOI: https://doi.org/10.1158/1535-7163.MCT-17-0575
- Primary Citation of Related Structures:
6B3S - PubMed Abstract:
Acquired resistance to cetuximab, an antibody that targets the EGFR, impacts clinical benefit in head and neck, and colorectal cancers. One of the mechanisms of resistance to cetuximab is the acquisition of mutations that map to the cetuximab epitope on EGFR and prevent drug binding. We find that necitumumab, another FDA-approved EGFR antibody, can bind to EGFR that harbors the most common cetuximab-resistant substitution, S468R (or S492R, depending on the amino acid numbering system). We determined an X-ray crystal structure to 2.8 Å resolution of the necitumumab Fab bound to an S468R variant of EGFR domain III. The arginine is accommodated in a large, preexisting cavity in the necitumumab paratope. We predict that this paratope shape will be permissive to other epitope substitutions, and show that necitumumab binds to most cetuximab- and panitumumab-resistant EGFR variants. We find that a simple computational approach can predict with high success which EGFR epitope substitutions abrogate antibody binding. This computational method will be valuable to determine whether necitumumab will bind to EGFR as new epitope resistance variants are identified. This method could also be useful for rapid evaluation of the effect on binding of alterations in other antibody/antigen interfaces. Together, these data suggest that necitumumab may be active in patients who are resistant to cetuximab or panitumumab through EGFR epitope mutation. Furthermore, our analysis leads us to speculate that antibodies with large paratope cavities may be less susceptible to resistance due to mutations mapping to the antigen epitope. Mol Cancer Ther; 17(2); 521-31. ©2017 AACR .
Organizational Affiliation:
Graduate Group in Biochemistry and Molecular Biophysics, University of Pennsylvania, Philadelphia, Pennsylvania.