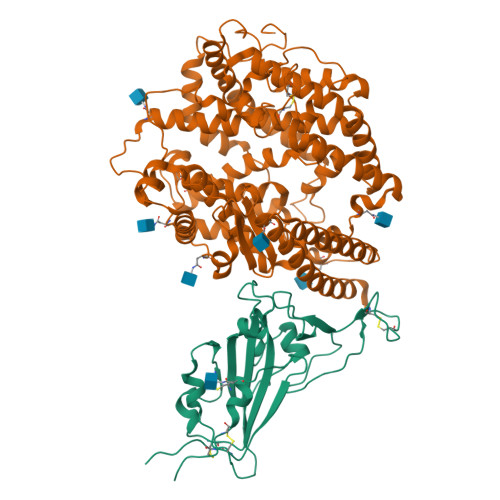
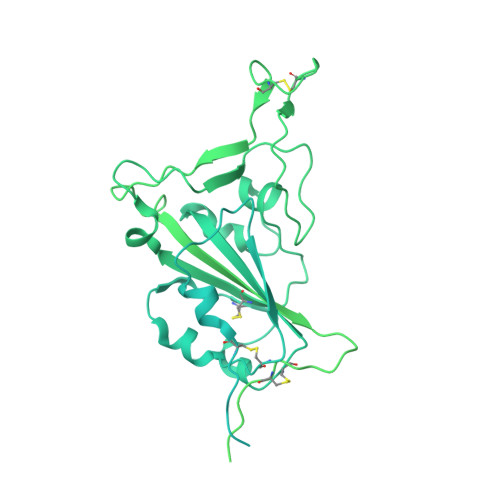
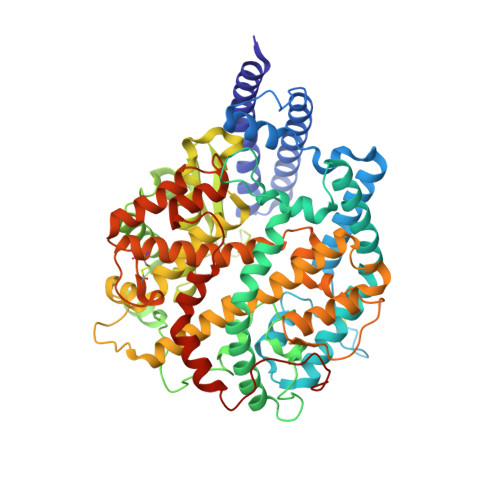
Structural analysis of receptor engagement and antigenic drift within the BA.2 spike protein.
Saville, J.W., Mannar, D., Zhu, X., Berezuk, A.M., Cholak, S., Tuttle, K.S., Vahdatihassani, F., Subramaniam, S.(2023) Cell Rep 42: 111964-111964
- PubMed: 36640338
- DOI: https://doi.org/10.1016/j.celrep.2022.111964
- Primary Citation of Related Structures:
8DM1, 8DM2, 8DM3, 8DM4, 8DM5, 8DM6, 8DM7, 8DM8, 8DM9, 8DMA - PubMed Abstract:
The BA.2 sub-lineage of the Omicron (B.1.1.529) severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) variant rapidly supplanted the original BA.1 sub-lineage in early 2022. Both lineages threatened the efficacy of vaccine-elicited antibodies and acquired increased binding to several mammalian ACE2 receptors. Cryoelectron microscopy (cryo-EM) analysis of the BA.2 spike (S) glycoprotein in complex with mouse ACE2 (mACE2) identifies BA.1- and BA.2-mutated residues Q493R, N501Y, and Y505H as complementing non-conserved residues between human and mouse ACE2, rationalizing the enhanced S protein-mACE2 interaction for Omicron variants. Cryo-EM structures of the BA.2 S-human ACE2 complex and of the extensively mutated BA.2 amino-terminal domain (NTD) reveal a dramatic reorganization of the highly antigenic N1 loop into a β-strand, providing an explanation for decreased binding of the BA.2 S protein to antibodies isolated from BA.1-convalescent patients. Our analysis reveals structural mechanisms underlying the antigenic drift in the rapidly evolving Omicron variant landscape.
Organizational Affiliation:
Department of Biochemistry and Molecular Biology, University of British Columbia, Vancouver, BC V6T 1Z3, Canada.