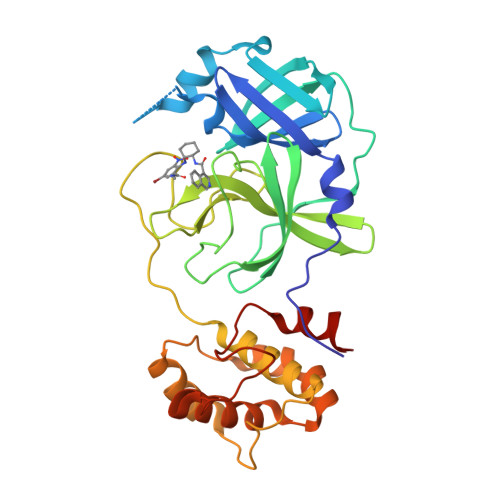
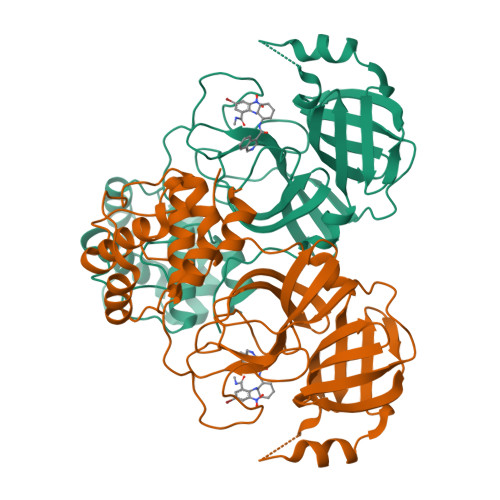
Resistance mechanisms of SARS-CoV-2 3CLpro to the non-covalent inhibitor WU-04.
Zhang, L., Xie, X., Luo, H., Qian, R., Yang, Y., Yu, H., Huang, J., Shi, P.Y., Hu, Q.(2024) Cell Discov 10: 40-40
- PubMed: 38594245
- DOI: https://doi.org/10.1038/s41421-024-00673-0
- Primary Citation of Related Structures:
8K67, 8K68, 8K6A, 8K6B, 8K6C, 8K6D - PubMed Abstract:
Drug resistance poses a significant challenge in the development of effective therapies against SARS-CoV-2. Here, we identified two double mutations, M49K/M165V and M49K/S301P, in the 3C-like protease (3CLpro) that confer resistance to a novel non-covalent inhibitor, WU-04, which is currently in phase III clinical trials (NCT06197217). Crystallographic analysis indicates that the M49K mutation destabilizes the WU-04-binding pocket, impacting the binding of WU-04 more significantly than the binding of 3CLpro substrates. The M165V mutation directly interferes with WU-04 binding. The S301P mutation, which is far from the WU-04-binding pocket, indirectly affects WU-04 binding by restricting the rotation of 3CLpro's C-terminal tail and impeding 3CLpro dimerization. We further explored 3CLpro mutations that confer resistance to two clinically used inhibitors: ensitrelvir and nirmatrelvir, and revealed a trade-off between the catalytic activity, thermostability, and drug resistance of 3CLpro. We found that mutations at the same residue (M49) can have distinct effects on the 3CLpro inhibitors, highlighting the importance of developing multiple antiviral agents with different skeletons for fighting SARS-CoV-2. These findings enhance our understanding of SARS-CoV-2 resistance mechanisms and inform the development of effective therapeutics.
Organizational Affiliation:
Zhejiang University, Hangzhou, Zhejiang, China.