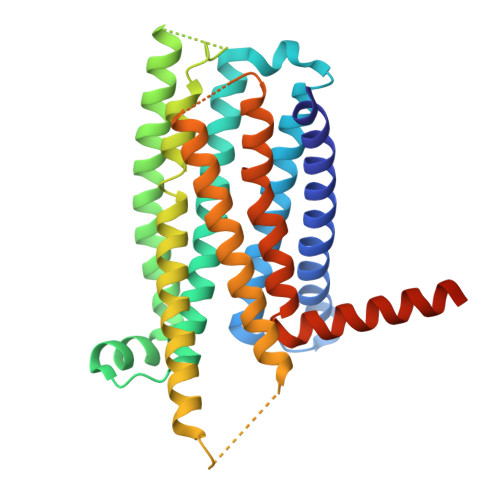
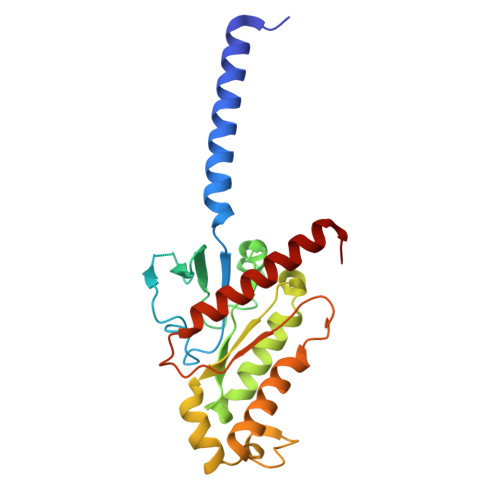
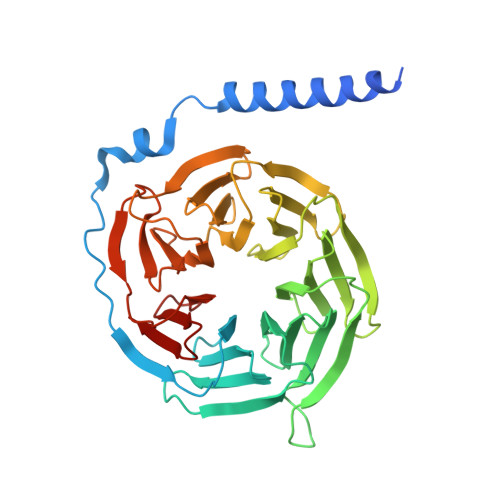
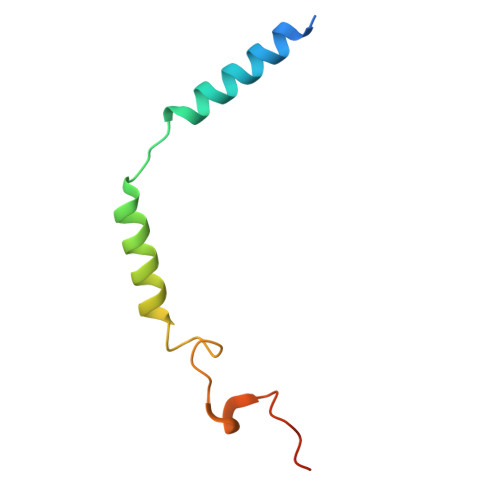
A bitter anti-inflammatory drug binds at two distinct sites of a human bitter taste GPCR.
Peri, L., Matzov, D., Huxley, D.R., Rainish, A., Fierro, F., Sapir, L., Pfeiffer, T., Waterloo, L., Hubner, H., Peleg, Y., Gmeiner, P., McCormick, P.J., Weikert, D., Niv, M.Y., Shalev-Benami, M.(2024) Nat Commun 15: 9991-9991
- PubMed: 39557861
- DOI: https://doi.org/10.1038/s41467-024-54157-6
- Primary Citation of Related Structures:
8RQL - PubMed Abstract:
Bitter taste receptors (TAS2Rs), a subfamily of G-protein coupled receptors (GPCRs) expressed orally and extraorally, elicit signaling in response to a large set of tastants. Among 25 functional TAS2Rs encoded in the human genome, TAS2R14 is the most promiscuous, and responds to hundreds of chemically diverse ligands. Here we present the cryo-electron microscopy (cryo-EM) structure of the human TAS2R14 in complex with its signaling partner gustducin, and bound to flufenamic acid (FFA), a clinically approved nonsteroidal anti-inflammatory drug. The structure reveals an unusual binding mode, where two copies of FFA are bound at distinct pockets: one at the canonical receptor site within the trans-membrane bundle, and the other in the intracellular facet, bridging the receptor with gustducin. Together with a pocket-specific BRET-based ligand binding assay, these results illuminate bitter taste signaling and provide tools for a site-targeted compound design.
Organizational Affiliation:
The Institute of Biochemistry, Food Science and Nutrition, Robert H. Smith Faculty of Agriculture, Food and Environment, The Hebrew University of Jerusalem, Rehovot, Israel.