Discovery of TNG908: A Selective, Brain Penetrant, MTA-Cooperative PRMT5 Inhibitor That Is Synthetically Lethal with MTAP -Deleted Cancers.
Cottrell, K.M., Briggs, K.J., Whittington, D.A., Jahic, H., Ali, J.A., Davis, C.B., Gong, S., Gotur, D., Gu, L., McCarren, P., Tonini, M.R., Tsai, A., Wilker, E.W., Yuan, H., Zhang, M., Zhang, W., Huang, A., Maxwell, J.P.(2024) J Med Chem 67: 6064-6080
- PubMed: 38595098
- DOI: https://doi.org/10.1021/acs.jmedchem.4c00133
- Primary Citation of Related Structures:
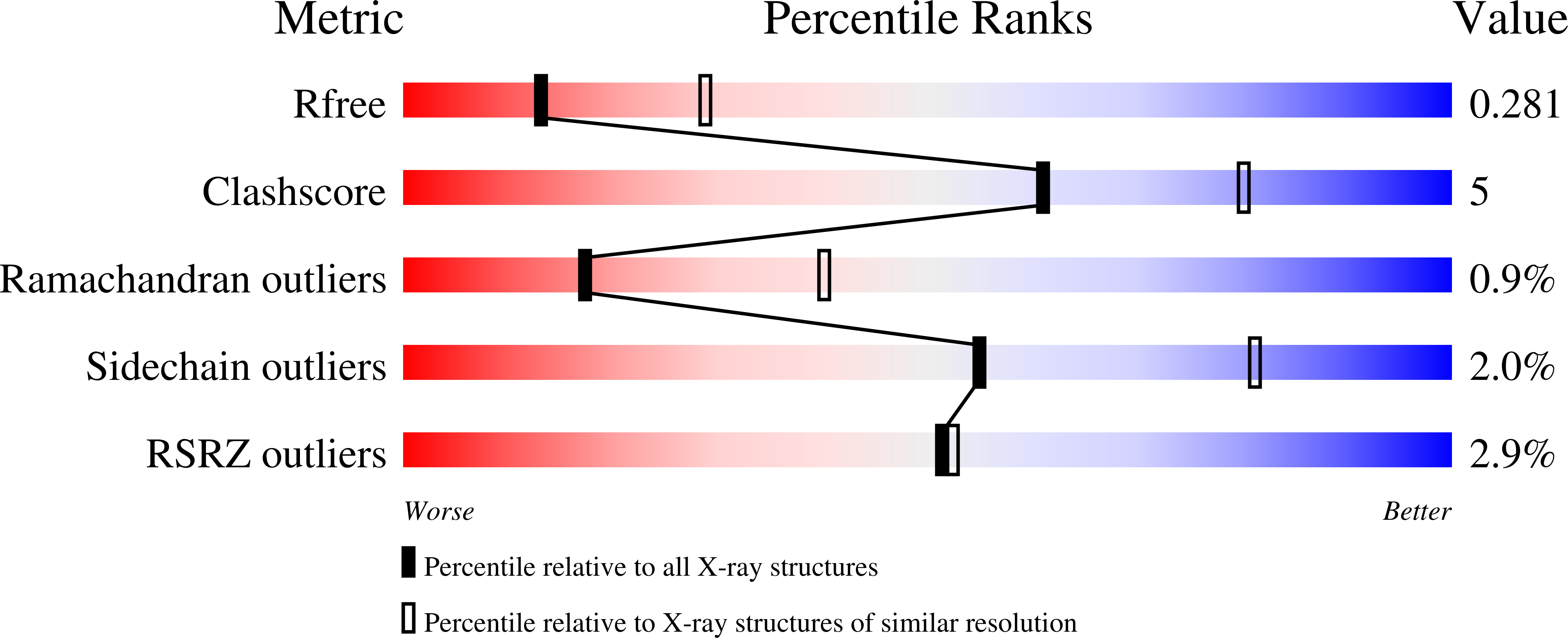
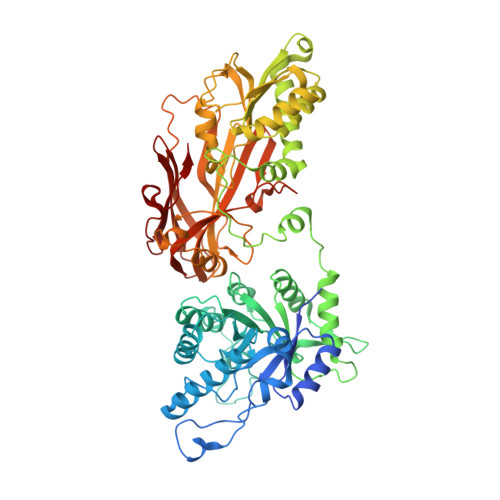
8VEO, 8VET, 8VEU, 8VEW, 8VEX, 8VEY - PubMed Abstract:
It has been shown that PRMT5 inhibition by small molecules can selectively kill cancer cells with homozygous deletion of the MTAP gene if the inhibitors can leverage the consequence of MTAP deletion, namely, accumulation of the MTAP substrate MTA. Herein, we describe the discovery of TNG908, a potent inhibitor that binds the PRMT5·MTA complex, leading to 15-fold-selective killing of MTAP -deleted (MTAP-null) cells compared to MTAP intact (MTAP WT) cells. TNG908 shows selective antitumor activity when dosed orally in mouse xenograft models, and its physicochemical properties are amenable for crossing the blood-brain barrier (BBB), supporting clinical study for the treatment of both CNS and non-CNS tumors with MTAP loss.
Organizational Affiliation:
Tango Therapeutics, Boston, Massachusetts 02215, United States.